| Posted: January 3, 2008 |
A crystal that Nature may have missed |
|
(Nanowerk News) For centuries, human beings have been entranced by the
captivating glimmer of the diamond. What accounts for the stunning
beauty of this most precious gem? As mathematician Toshikazu Sunada
explains in an article appearing today in the Notices of the American
Mathematical Society, some secrets of the diamond's beauty can be
uncovered by a mathematical analysis of its microscopic crystal
structure. It turns out that this structure has some very special,
and especially symmetric, properties. In fact, as Sunada discovered,
out of an infinite universe of mathematical crystals, only one other
shares these properties with the diamond, a crystal that he calls the
"K4 crystal". It is not known whether the K4 crystal exists in
nature or could be synthesized.
|
|
One can create an idealized mathematical model of a crystal by
focusing on its main features, namely, the atoms and the bonds between
them. The atoms are represented by points, which we will call
"vertices", and the bonds are represented as lines, which we will call
"edges". This kind of network of vertices and edges is called a
"graph". A crystal is built up by starting with a building-block
graph and joining together copies of itself in a periodic fashion.
Thus there are two patterns operating in a crystal: The pattern of
edges connecting vertices in the building-block graphs (that is, the
pattern of bonding relations between the atoms), and the periodic
pattern joining the copies of the graphs. One can create infinitely
many mathematical crystals this way, by varying the graphs and by
varying the way they are joined periodically.
|
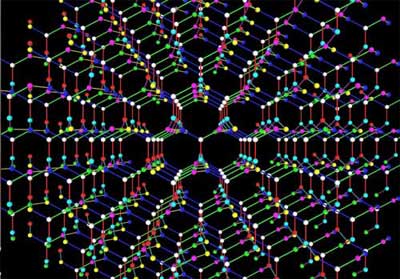 |
| K4 crystal. Created by Hisashi Naito.
|
|
The diamond crystal has two key properties that distinguish it from
other crystals. The first, called "maximal symmetry", concerns the
symmetry of the arrangement of the building-block graphs. Some
arrangements have more symmetry than others, and if one starts with
any given arrangement, one can deform it, while maintaining
periodicity and the bonding relations between the atoms, to make it
more symmetrical. For the diamond crystal, it turns out that no
deformation of the periodic arrangement can make it any more
symmetrical than it is. As Sunada puts it, the diamond crystal has
maximal symmetry.
|
|
Any crystal can be deformed into a crystal with maximal symmetry, so
that property alone does not distinguish the diamond crystal. But the
diamond crystal has a second special property, called "the strong
isotropic property". This property resembles the rotational symmetry
that characterizes the circle and the sphere: No matter how you rotate
a circle or a sphere, it always looks the same. The diamond crystal
has a similar property, in that the crystal looks the same when viewed
from the direction of any edge. Rotate the diamond crystal from the
direction of one edge to the direction of a different edge, and it
will look the same.
|
|
It turns out that, out of all the crystals that are possible to
construct mathematically, just one shares with the diamond these two
properties. Sunada calls this the K4 crystal, because it is made out
of a graph called K4, which consists of 4 points, in which any two
vertices are connected by an edge.
|
|
"The K4 crystal looks no less beautiful than the diamond crystal,"
Sunada writes. "Its artistic structure has intrigued me for some
time." He notes that, although the K4 crystal presently exists only
as a mathematical object, it is tempting to wonder whether it might
occur in nature or could be synthesized. This is not so far-fetched
as it may sound: The Fullerene, which has the structure of a soccer
ball (technically called a truncated icosahedron), was identified as a
mathematical object before it was found, in 1990, to occur in nature
as the C60 molecule.
|
|
Sunada's article, "Crystals That Nature Might Miss Creating", is
appearing in the February 2008 issue of the AMS Notices and is
being posted online today at http://www.ams.org/notices.
|

