| Jul 02, 2013 |
Universal bound states of fermions
|
|
(Nanowerk News) The behaviour of a few particles can have important ramifications for how a system of many particles behaves. In particular, bound states of two, three or more particles can determine how particles will cluster, i.e. how particles are correlated, in a gas or fluid. Examples include paired superfluids in dilute atomic vapours, and the clustering of nucleons in a nucleus. Indeed, for short-range interactions between particles, there is the prospect of universal correlations that are insensitive to the microscopic details and are thus relevant to a wide range of systems.
|
|
Now, Dr Meera Parish, from the London Centre for Nanotechnology, and Dr Jesper Levinsen have discovered a universal bound state of four atoms (i.e. a tetramer) in two dimensions ("Bound States in a Quasi-Two-Dimensional Fermi Gas").
|
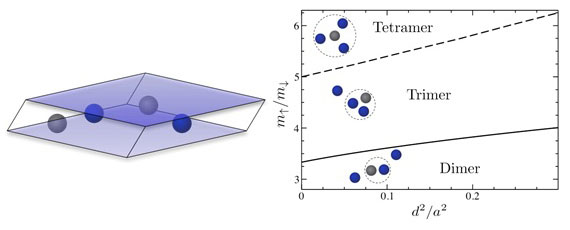 |
| (left) Two species (,¯) of fermionic atom are confined in a two-dimensional layer of width d. (right) Bound states of three or more atoms can form above a critical mass ratio as the confinement width is varied.
|
|
Such bound states have been known to exist for bosonic atoms, which typically like to cluster, but Parish and Levinsen are the first to demonstrate the existence of a universal tetramer entirely composed of fermionic atoms. Fermions are famous for avoiding one another, with the so-called exclusion principle being responsible for the structure of the periodic table and the stability of matter. However, fermions may bind together if there is a lighter particle that dances around and effectively mediates an attraction between them, thus resulting in a tetramer of three heavy fermions and one light.
|
|
In principle, such a tetramer can be realised in experiment by confining atoms, such as potassium and lithium, to a two-dimensional layer with lasers. Varying the confinement can then lead to the formation of larger and larger clusters of fermions. Such a set-up could be used to tune the effective interactions and correlations in a system of many atoms, and this could ultimately be used to simulate other more complicated materials.
|

