| Aug 31, 2013 |
Nanosized aspirin increases effectiveness
|
|
(Nanowerk News) Aspirin has enjoyed its medicinal popularity for over 110 years. The therapeutic usage of aspirin in diverse indications, such as acute inflammation and pain, cardiovascular diseases, stroke, pregnancy complications, cancer, diabetes, and Alzheimer’s disease, reflects its colorful achievements.
|
|
However, clinical trials have also exposed the limitations of aspirin therapy. The major problem is implicated in the resistance or nonresponse to aspirin. Aspirin resistance and nonresponse are defined as the incomplete inhibition of platelet function of the patients treated with aspirin, even though the patients with a specific disease receive a clinically effective dosage.
|
|
This phenomenon severely decreases the therapeutic efficacy of aspirin in the treatment of various diseases including type 2 diabetes, asthma, cardiovascular disease, stroke, coronary artery disease, and pediatric cardiac surgery.
|
|
A series of efforts have been made in order to overcome the clinical shortcomings of aspirin. For example, to increase the platelet sensitivity of diabetes mellitus patients, excellent near-normal metabolic control was clarified and the dosing schedules of aspirin were altered; to increase the platelet sensitivity of cardiovascular patients having elevated plasma cholesterol, aspirin was combined with an inhibitor of lipid peroxidation; to increase the platelet sensitivity of cardiovascular patients at risk for secondary atherothrombosis, aspirin was combined with a statin; to increase the sensitivity of colon cancer, aspirin was covalently linked to nontoxic well-defined 3-hydroxybutanoic acid oligomers; to avoid aspirin intolerance, patients were recommended to use alternative drugs; and to enhance the safety of oral aspirin challenge in patients with aspirin-exacerbated respiratory disease, pretreatment with leukotriene-modified drugs was recommended. In spite of some achievements made by these efforts, resistance and nonresponse remains the major obstacle for the utility of aspirin in the treatment of various diseases including cardiovascular diseases.
|
|
Aiming at avoiding resistance and nonresponse to aspirin, a new study by researchers in China, reported in ACS Nano ("Nanosized Aspirin-Arg-Gly-Asp-Val: Delivery of Aspirin to Thrombus by the Target Carrier Arg-Gly-Asp-Val Tetrapeptide") tested a hypothesis that RGDV covalently modified aspirin, aspirin-Arg-Gly-Asp-Val (A-RGDV), should be stable in the circulation, release aspirin in thrombi, and inhibit the formation of thrombi.
|
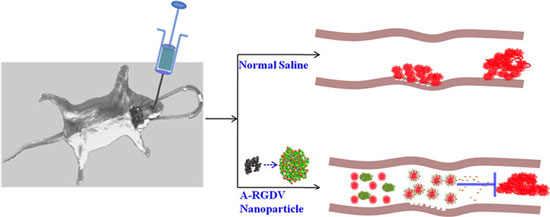 |
| The delivery investigation implies that in rat blood A-RGDV is able to keep its molecular integrity, while in a thrombus it releases aspirin. The in vitro antiplatelet aggregation assay suggests that A-RGDV selectively inhibits arachidonic acid induced platelet aggregation. The mechanisms of action probably include releasing aspirin, modifying cyclic oxidase, and decreasing the expression of GPIIb/IIIa. The in vivo assay demonstrates that the effective antithrombotic dose of A-RGDV is 16700-fold lower than the nonresponsive dose of aspirin. (© ACS)
|
|
In contrast to the nanoparticles that consisted of macromolecules such as biomaterials, engineered materials, and pharmaceutical materials, the present nanoparticles consisted of a small-molecule, aspirin-linked tetrapeptide. In normal blood vessels the nanoparticles were optionally delivered in the blood without interfering with the integrated nanostructure and molecular structure. If the vascular endothelium is injured and the platelets are activated, the nanoparticles will target the injured site, interact with the activated platelets, bind the receptor GPIIb/IIIa, release aspirin, and block the conversion of AA into PGG2. Thereby, A-RGDV functions as a delivery system of aspirin, and RGDV functions as the target carrier of aspirin and miraculously increases the efficacy of aspirin in inhibiting thrombosis. Thus, the present paper provides a strategy of RGD peptide carrying aspirin to the thrombus, releasing aspirin inside the thrombus, and finally avoiding aspirin resistance and nonresponse.
|

