| Posted: Jul 15, 2015 |
Quantum dots light the way
|
|
(Nanowerk News) Polymer nanoparticles that release medicine at controlled rates inside cells have the potential to enhance the efficacy of many clinical drugs. A*STAR researchers have now developed an eye-catching way to evaluate the performance of different polymer drug-delivery formulations using luminescent quantum dots as imaging labels ("Evaluation of Polymeric Nanoparticle Formulations by Effective Imaging and Quantitation of Cellular Uptake for Controlled Delivery of Doxorubicin").
|
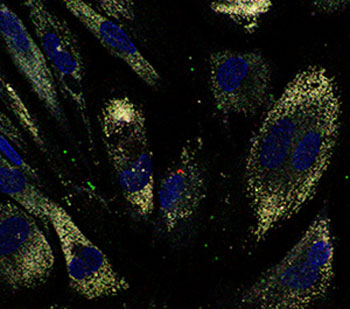 |
| Confocal images of quantum dots localized within colon cells can guide the development of innovative drug delivery formulations. (© Wiley-VCH Verlag)
|
|
Tiny, inorganic quantum-dot crystals are finding increasing use as biological probes due to their powerful optical characteristics. By stimulating the dots with laser light, researchers can obtain sharp images to monitor processes such as drug delivery for much longer time frames than nearly any other technique. However, a key challenge lies in incorporating hydrophobic quantum dots into biocompatible, water-soluble polymers.
|
|
Ming-Yong Han and co-workers from the A*STAR Institute of Materials Research and Engineering in Singapore turned to a copolymer known as poly(D,L-lactide-co-glycolide), or PLGA, for their quantum-dot imaging strategy. This non-toxic material has tunable water-repelling or water-attracting ability, depending on the proportion of lactic and glycolic acid components. It is also an ideal drug delivery platform for the popular anticancer drug doxorubicin — a fluorescent molecule used to treat diseases including leukemia and Hodgkin’s lymphoma.
|
|
“The choice of polymer and nanoparticle preparations plays an important role in making uniformly fluorescent particles,” says co-author Choon Peng Teng. “Different hydrophobic or hydrophilic interactions affect how quantum dots are incorporated.”
|
|
The team synthesized two kinds of PLGA nanoparticles — one loaded with doxorubicin, and the other containing quantum-dot bio-labels — and incubated them in a culture of human colon cells. After two hours, confocal imaging revealed that both kinds of polymer nanoparticles were engulfed by the cells through an endocytosis mechanism and internalized into the cytoplasm (see image). The bright emissions from the dots enabled the researchers to quantify the uptake as 25 per cent of the cell volume.
|
|
Since the behavior of the quantum dot-labeled nanoparticles paralleled the doxorubicin-impregnated materials, Han and colleagues realized this imaging system could model the effectiveness of other important drug-delivery schemes. Initial investigations appear promising — the quantum-dot-loaded PLGA nanoparticles mimicked different drug-delivery systems for targeting brain, lung and breast cancer cell lines, and were compatible with both water-soluble and water-insoluble drugs.
|
|
One further advantage of this approach, notes co-author Khin Yin Win, is that it can simulate the action of non-fluorescent anticancer drugs previously untraceable with confocal imaging. “This model can facilitate monitoring biocompatibility and cellular uptake, but it can also evaluate how feasible certain materials are as drug carriers,” she says. “This could lead to more innovative drug-delivery systems.”
|

