| Jul 14, 2023 |
Space has gotten small with metallic, planet-like nanodroplets
(Nanowerk News) Liquid metal, planet-like nanodroplets are successfully formed with a new technique developed at RMIT University, Australia.
|
|
Like our own Planet Earth, the nanodroplets feature an outer ‘crust’, a liquid metal ‘mantle’, and a solid ‘core’.
|
|
The solid intermetallic core is the key to achieving a more homogenous mix, ‘locking up’ the same amount of solute (ie, the ‘target’ metals) in each alloyed droplet.
|
|
The research team achieved homogeneity via complete dissolution within the liquid-metal media, made possible by high-temperature molten salt.
|
|
The discovery creates new research opportunities in fundamental liquid-metal chemistry as well as applications as diverse as flexible electronics, phase-change materials, catalysts and fuel cells, and silver-based antimicrobials.
|
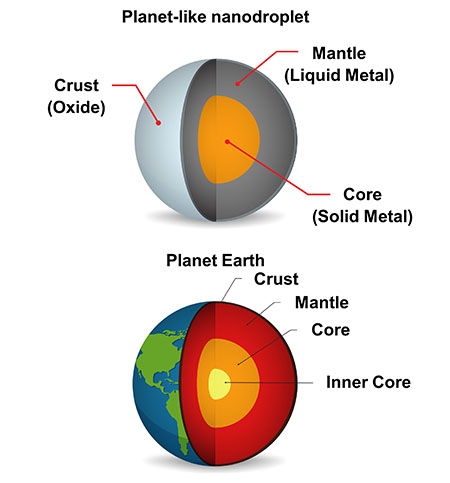 |
| Planet-like nanodroplets have an outer (oxide) shell, liquid (metal) mantle and suspended, solid central core (intermetallic). (Image: FLEET)
|
Liquid metal nanodroplets shake apart
|
|
Liquid metals have emerged as a promising new frontier of chemical research in recent years, acting as a novel reaction interface for solvents and catalysts.
|
|
They can also act as a functional material offering high conductivity, due to delocalised metallic bonds, and a soft, fluid interior.
|
|
With emerging catalysis, sensing and nano-electronic applications relying on achieving large surface areas, synthesis of liquid metal nanodroplets has become an important focus.
|
|
There are many combinations possible when alloying for specific applications, for example dissolving copper (the solute) in liquid gallium (the metallic solvent).
|
|
The liquid-metal nanodroplets are created by mechanical agitation using sound waves in a solvent such as ethanol or water.
|
|
However, during this ‘sonication’ process, liquid-metal alloys have tended to ‘de-alloy’, ie, to break apart into their constituent metals.
|
|
This is a result of previous methods attempting to dissolve the metals at relatively low temperatures, near room temperature. “Just as it’s possible to dissolve more sugar in warm water than in cold water, more copper can be dissolved in warmer gallium,” says lead author Caiden Parker, a PhD candidate at RMIT.
|
|
At low temperatures, some of the solute metal re-forms into larger, solid particles before complete dissolution.
|
|
The resulting composition has inconsistent, inhomogeneous properties, with the composition of individual nanodroplets varying considerably. “In extreme cases, many or even most nanodroplets may be essentially devoid of the solute metal, which ends up being concentrated in only very few particles,” says corresponding author Dr Torben Daeneke, also at RMIT.
|
|
This inhomogeneity and the presence of intermetallic compounds poses considerable difficulties for researchers wishing to understand the fundamental mechanisms at work in liquid metal chemistry.
|
High-temperatures and salts form homogenous, planet-like nanodroplets
|
|
“The core is the key!”
|
|
In the new study (Advanced Functional Materials, "Synthesis of planet-like liquid metal nanodroplets with promising properties for catalysis"), RMIT researchers resolved the problem of dealloying by significantly heating the synthesis process (as high as 400°C) to ensure the solute metal is completely dissolved and introducing a carefully selected molten-salt suspension fluid.
|
|
Sodium acetate was selected because it remains stable at high temperature and can be easily removed afterwards.
|
|
The resulting nanodroplets feature an interesting ‘planet-like’ structure consisting of an outer (oxide) shell, a liquid (metal) mantle and a suspended, solid central core (intermetallic).
|
|
“We were immediately struck by the nanodroplets’ similarity to an Earth-like planet, with a solid outer shell, a liquid metal mantle, and a solid metal core,” says Caiden.
|
|
That solid core is the key to the success of the new technique, ‘locking up’ the same amount of solute in each alloyed droplet.
|
|
“We were also delighted to see that our new metallic planet-like nanodroplets were everywhere!” continues Caiden.
|
|
The system was homogenously spread, with output yield significantly improved. Transmission Electron Microscope (TEM) analysis confirmed the core structure is observed in almost every droplet.
|
|
The presence of the solid core also promotes a very interesting use for the planet-like nanodroplets in catalytic reactions, ‘speeding up’ chemical reactions.
|
|
The copper-gallium nanodroplets studied provided promising results in electrocatalytic oxidation of ethanol, which could be applied in ethanol fuel cells.
|
|
Removal of the sodium acetate is important prior to this catalytic reaction, with the salt easily cleaned away in simple water baths.
|
What’s next?
|
|
The promising new technique opens up the potential use of high-surface area nanodroplets in a wide range of future applications, including, but not limited to, electronics or catalytic materials.
|
|
The physical scale of the nanodroplets (ie, nano rather than micro) will also assist fundamental studies of liquid-metal chemistry, including looking into the precise nature of bond formation within liquid metals, solvation capabilities, crystallization dynamics and the general colloidal chemistry that may occur within various molten metal systems.
|
|
“The planet-like structures are like little miniature laboratories, allowing us to study how molten metals behave at an atomic level,” says Torben.
|
|
While the study proved viability of the new technique using a copper-gallium system, the authors expect further work to confirm that the technique will be successful using other combinations of solute and solvent alloy systems, beginning with silver, zinc, or bismuth in liquid gallium, tin or indium.
|
|
“A key advantage of liquid-metal systems is the ability to adjust the metal mix for certain applications, dependent on the properties of the constituent metals,” says Caiden.
|
|
“For example, copper is a great electrical conductor. When we combine copper with gallium, we not only save significant cost in material consumption, but also open the way to flexible electronics, such as what you might have seen in sci-fi movies.”
|
|
Potentially, copper can also be utilised for its thermal properties, with potential application of copper-based nanodroplets in heat dissipation systems.
|
|
Nanodroplet catalysis applications based on the ability of copper to speed up reactions has already been tested in the new study, with improved active-site area in addition to material synthesis savings.
|
|
Looking at another metal, silver has previously found applications based on its anti-microbial properties, and once combined with gallium could create a more bioavailable alternative.
|
|
“Thus the potential applications of the new technology are extremely wide. Any industries in need of nanomaterials can utilise the system, with constituent metals varying according to application,” says Torben.
|

