| Sep 26, 2023 |
Thiol ligands modify metal nanocluster structures and optical properties
(Nanowerk News) Metal nanoclusters are tiny, crystalline structures up to two nanometers (2 x 10-9 meters) in diameter that contain a few to hundreds of metal atoms. Understanding the precise assembly of metal nanoclusters is paramount to determining how different structures affect the properties and molecular interactions of these materials. Researchers recently synthesized two similar gold-silver (Au9Ag6) nanoclusters in a highly controlled manner to determine the precise atomic structure of each nanocluster and the effects of specific thiol ligands, or sulfur-containing binding molecules, on material synthesis.
|
|
Given their extremely small size, metal nanoclusters have unique properties and potential applications in nanomedicine, chemical engineering and quantum mechanics. Chemists from Anhui University recently used two different thiol ligands, SPhpOMe and SPhoMe, to determine how each ligand would affect Au9Ag6 nanocluster synthesis. Remarkably, depending on the thiol ligand used, the nanoclusters formed different higher-order superlattice structures in which different conformations of the material repeated in the structure. In this case, the thiol ligand was responsible for creating either an ABAB (for the Au9Ag6-SPhpOMe nanocluster) or an ABCDABCD (for the Au9Ag6-SPhoMe nanocluster) superlattice structure pattern depending on which thiol ligand was used.
|
|
The team published their results in Polyoxometalates ("Ligand-correlated crystalline assembly of nanoclusters with atomic precision"), published by Tsinghua University Press.
|
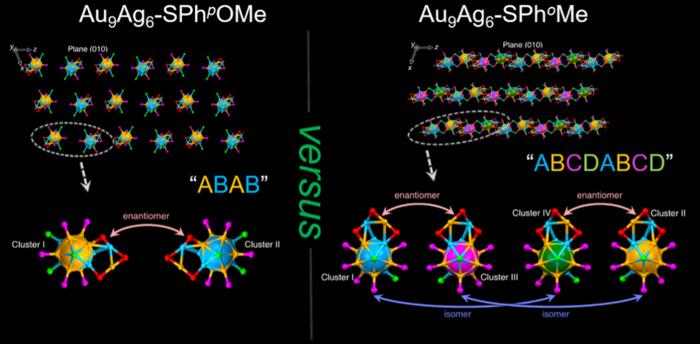 |
| Two thiol ligands, SPhpOMe and SPhoMe, produce chiral, mirror-image gold-silver (Au9Ag6) nanocluster structures depending on the thiol ligand used during assembly. Clusters created using the SPhpOMe ligand form two variants that arrange in an ABAB higher-order superlattice pattern and clusters formed using the SPhoMe ligand form four variants that assemble in an ABCDABCD pattern. (Image: Polyoxometalates, Tsinghua University Press)
|
|
“[T]he highest level of knowledge in Nanoscience [is] atomic precision. [This is] why… structural science is [so important] in Nanoscience [and] other [fields like] Structural Chemistry and Structural Biology. [By] studying the assembly pattern of metal nanoclusters [with] atomic precision, [we gain] the most essential knowledge [of] [molecular and supramolecular] structure evolutions… [and] structure-property correlations,” said Xi Kang, an author of the paper and researcher in the Department of Chemistry and Centre for Atomic Engineering of Advanced Materials at Anhui University in Anhui, China.
|
|
The team used single-crystal X-ray diffraction (SC-XRD) and electrospray ionization mass spectrometry (ESI-MS) to verify the exact structure of each synthesized gold-silver nanocluster, using either SPhpOMe or SPhoMe as a thiol ligand. Interestingly, the thiol ligand used during synthesis changed the packing of gold and silver atoms within the core of the nanocluster and not just the outer nanocluster structure. The data suggested a more contracted structure for the SPhoMe-ligand gold-silver nanocluster (Au9Ag6-SPhoMe) compared to the SPhpOMe-ligand nanocluster (Au9Ag6-SPhpOMe).
|
|
The research team also noted that metal-metal bond lengths were responsible for the additional Au9Ag6-SPhoMe structural variants (ABCD) compared to the Au9Ag6-SPhpOMe (AB) nanoclusters.
|
|
The differing molecular structures between the Au9Ag6-SPhoMe and Au9Ag6-SPhpOMe nanoclusters changed the superlattice structures of the materials, as well as their optical properties. Initially the team found that the optical absorptions of the two materials were similar, indicating the nanoclusters possessed similar frameworks and electron configurations.
|
|
In contrast, the photoluminescence intensity of Au9Ag6-SPhoMe nanoclusters at 795 nm and 785 nm nm wavelengths of light were greater than Au9Ag6-SPhpOMe nanoclusters (795 nm and 758 nm) in solution and crystalline state, respectively. The authors attributed these optical property changes to the increased noncovalent binding interactions in the Au9Ag6-SPhoMe nanocluster structure, or distinct combinations of the electronic coupling and the lattice-origin, non-radiative decay pathways occurring through electron-phonon interactions for two nanoclusters.
|
|
“This work not only reveals two nanoclusters displaying dramatically different arrangements in their crystal units due to the strong ligand effect, but also highlights that… ligand engineering should be an effective strategy for designing highly ordered cluster-based assemblies with customized structures and performances,” said Kang.
|
|
With this improved understanding of thiol ligand effects on nanocluster assembly, the research team looks forward to applying this knowledge to create new nanoclusters with different structures and properties. “[T]he study of nanoclusters should head for its next step: the practical application. We hope the findings in this work… lay a foundation for the fabrication of cluster-based assembled nanomaterials with high application values. Future works will focus on promoting the ligand engineering strategy to cluster-based assembled nanomaterials and further promoting their applications in different fields, especially… optics,” said Kang.
|

