| Posted: August 19, 2009 |
Molten proteins - surface-modified liquid protein with liquid-crystalline properties |
|
(Nanowerk News) Proteins are solids. When heated they do not melt; instead, they decompose or sublime directly to the gas phase at low pressures. They cannot be converted into a liquid form unless they are dissolved in a solvent. A team at the University of Bristol (UK) and the Max Planck Institute of Colloids and Interfaces in Golm (Germany) has now successfully liquefied a protein without the assistance of a solvent. As the research team headed by Stephen Mann reports in the journal Angewandte Chemie (Solvent-Free Protein Liquids and Liquid Crystals), the trick is to modify the surface of the protein with a polymeric surfactant.
|
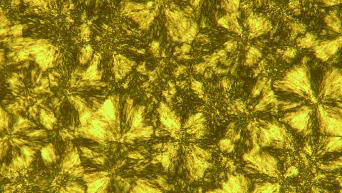 |
| Surface-modified liquid protein with liquid-crystalline properties.
|
|
The researchers used ferritin for their experiments. This large protein serves animals and plants as a storage material for iron. Ferritin forms a hollow sphere that can hold thousands of iron ions. Adam Perriman, a researcher in the Mann lab, attached polymer chains consisting of a polyethylene oxide portion and a hydrocarbon portion to these iron-containing ferritin spheres. About 240 polymer chains were attached to every ferritin molecule. A solution of proteins modified in this way was freeze-dried. |
|
The resulting dry powder could be melted to form a transparent, viscous red liquid that solidified only upon cooling to –50 °C. In the temperature range between 30 and 37 °C the modified protein is in a liquid-crystalline state, which means the molecules are oriented more or less uniformly but (at least partly) lack the three-dimensional lattice that is formed in the crystalline state. At higher temperatures, the modified protein acts like a normal liquid. It only decomposes at temperatures above 400 °C.
|
|
|
|
How does the liquefaction work? The surfactant chains on the ferritin surface keep the protein spheres apart and shield their surfaces. This prevents the electrostatic attractive forces between polar molecular groups of neighboring spheres from holding the proteins together in a solid. The spheres are instead held together by attractive forces between the hydrocarbon ends of the surfactant chains. These forces are only strong enough to hold the molecules together as a liquid. Between 30 and 37 °C the surfactant chains arrange themselves in an ordered pattern, giving the substance liquid-crystalline properties.
|
|
“This is a very exciting result with fundamental significance for understanding liquids comprising nanostructured components,” says Mann. “Also, it represents a possible way forward to a novel state of biomolecular matter, and could therefore have a number of important applications, for example in biomedical and sensor technology.”
|

