| Jun 22, 2018 |
Forever young catalyst reduces diesel emissions
|
|
(Nanowerk News) Diesel engines release pollutants. Specially designed catalysts reduce the amount of pollutants released. But what causes such catalysts to fail? Atom probe tomography provides the first direct observations of the problems (Nature Communications, "Nanoscale tomography reveals the deactivation of automotive copper-exchanged zeolite catalysts").
|
|
Scientists saw the atom-level redistributions that deactivate catalysts during simulated exposure to 135,000 miles of diesel exhaust. The team analyzed two copper-exchanged zeolite catalysts. The catalyst Cu-SSZ-13 has superior performance and stability compared to other commercial zeolite catalysts.
|
|
The more common industrial catalyst, Cu-ZSM-5, can’t remain active for a vehicle’s lifetime. In this work, loss of catalytic activity correlates with the formation of tiny clusters. The clusters result from the collapse of the Cu-ZSM-5 framework and copper migration.
|
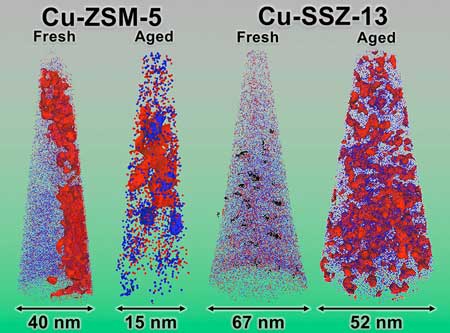 |
| Red and blue represent positions of copper and aluminum atoms, respectively, for two zeolite catalysts (Cu-SSZ-13 and Cu-ZSM-5) used in diesel-vehicle catalytic converters. The catalysts are fresh (that is, not exposed to diesel engine exhaust) or aged (simulated exposure to 135,000 miles of diesel engine exhaust). After long-term aging, the Cu-SSZ-13 catalyst still cleans exhaust while Cu-ZSM-5 does not perform as well. Atom-by-atom 3-D image reconstructions reveal that the degradation of Cu-ZSM-5 is due to severe aggregation of copper and aluminum to form a deleterious phase, CuAl2O4. (Image: Oak Ridge National Laboratory)
|
|
Gaining a complete picture of the mechanism of catalyst deactivation through atom-by-atom 3-D reconstruction provides key information that the automotive industry needs to design better catalysts. These catalysts will perform as well at the end of a vehicle’s life as they did the day the car left the factory. Such performance is vital to keeping our air clean.
|
|
In the chemical industry, where zeolite catalysts are broadly used, the knowledge gained may advance sustainable processes. These processes include biomass conversion, green plastics production, capture and conversion of carbon dioxide, remediation of air pollution and water purification.
|
|
Decreasing fuel consumption and emissions in diesel vehicles requires end-of-tailpipe technologies. To reduce emissions of nitrogen oxide pollutants that cause the formation of acid rain and smog, the most effective strategy is ammonia selective catalytic reduction using urea as the source of the ammonia reductant with the reaction occurring over a copper-exchanged zeolite catalyst.
|
|
Such a catalyst is Cu-ZSM-5, which was originally described in 1986, but could not be fully commercialized as it deactivates well before the end of vehicle life. A major breakthrough in zeolite technology occurred in the mid-2000s when another copper-exchanged zeolite catalyst, Cu-SSZ-13, was discovered with unsurpassed activity and stability (that is,it cleans exhaust better over a longer period of time), and commercialization commenced quickly in 2010.
|
|
Significant gaps remain in understanding the deactivation of zeolite catalysts, and thus, researchers at Utrecht University and Oak Ridge National Laboratory used APT to peer inside industry-relevant zeolites to visualize their nanoscale, elemental structure in 3-D. They looked at both Cu-ZSM-5 and Cu-SSZ-13 catalysts in fresh (that is, not exposed to diesel engine exhaust) and aged (simulated exposure to 135,000 miles of diesel exhaust) conditions.
|
|
The study showed the nanoscale formation of a culprit phase causing catalytic deactivation (CuAl2O4l) through copper-aluminum aggregation, which was severe in Cu-ZSM-5. In contrast, Cu-SSZ-13 did not exhibit severe copper-aluminum aggregation nor evidence of the deactivating CuAl2O4 phase and was still able to optimally clean combustion products.
|

