| Dec 03, 2013 |
Researchers make progress in fabricating three-dimensional metallic carbon
|
|
(Nanowerk News) Design and synthesis of three-dimensional (3D) metallic carbon that is stable under ambient conditions has been a long-standing dream. Recently a new progress has been made by Professor Wang Qian’s research group at the Center for Applied Physics and Technology (CAPT), College of Engineering of Peking University and her collaborators in this subject. They predict the existence of such phases consisting of interlocking hexagons. The findings are published online in the Early Edition of the Proceedings of the National Academy of Sciences on Nov. 4, 2013 ("Stable three-dimensional metallic carbon with interlocking hexagons").
|
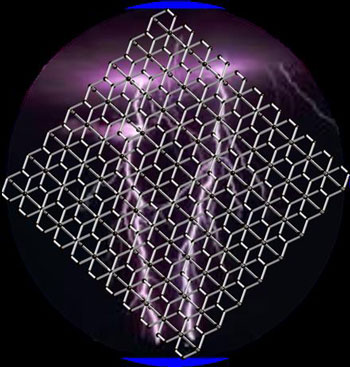 |
| Three-dimensional metallic carbon with interlocking hexagons.
|
|
Carbon is an amazing material: it not only forms the chemical basis for all known life but also because of its rich physics and chemistry, it displays an array of structures – from the age-old graphite and diamond to more recent C60 fullerene, one-dimensional nanotube, and two-dimensional graphene, and it takes a leading role in science and technology. However, synthesizing three-dimensional forms of metallic carbon that are stable under ambient conditions had long eluded researchers.
|
|
Using state-of-the-art theoretical calculations Wang and co-workers discovered that a form of carbon resembling interlocked versions of the hexagonal rings is metallic as well as dynamically, mechanically and thermally stable, and it may be synthesized chemically by using benzene or polyacenes molecules.
|
|
“We knew that metallicity in carbon structures arises due to sp2 bonding that leads to planar hexagonal structures while strength originates from sp3 bonding that leads to three-dimensional structures. Thus having both structures through interlocking hexagons seemed to combine the best of both worlds,” Wang said. Since the predicted structure is porous, it could have potential applications in catalysis for energy conversion and storage. Because of its light weight it may also find applications in space.
|

