| Posted: Jul 02, 2009 | |
Fluorescent gold nanoprobes for real-time detection of reactive oxygen species |
|
| (Nanowerk Spotlight) Environmental and behavioral factors such as excessive alcohol consumption, exposure to toxins and drugs, smoking and lack of sleep, may lead the body to produce superoxide radicals known as reactive oxygen species (ROS) that could cause cell damage through oxidation. Oxidative stress from ROS is implicated in aging and most diseases including cancer, heart disease, liver fibrosis, neurodegenerative diseases, autoimmune disorders. An excess of these reactive molecules can lead to oxidative stress and cellular damage, and toxicologists have identified ROS generation as a likely mechanism of nanoparticle toxicity. | |
| Since ROS plays an important role in various pathogenic processes, it has been recognized as an early indicator for cytotoxic events and cellular disorders. However, conventional chemical ROS probes have not fulfilled the rising need of in vitro and in vivo analysis of ROS generation due to auto-oxidation problems and poor specificity and sensitivity. | |
| Scientists in South Korea have now demonstrated a novel ROS-sensitive gold nanoprobe prepared from bio-inspired immobilization of fluorescein-labeled hyaluronic acid onto the surface of gold nanoparticles. This probe is highly stable under exposure to natural light and laser sources and extremely sensitive and specific to certain oxygen species. | |
| "Inorganic nanoparticle based signal-amplification and fluorescence energy transfer techniques have revolutionized the fields of molecular recognition, bioassay, and biosensing," Hyukjin Lee explains to Nanowerk. "The fluorescent gold nanoprobes that we have developed highlight the future possibilities of gold nanoparticle based imaging probes that can exceed conventional chemical imaging probes. Our study includes the interdiciplinary field of inorganic materials (gold nanoparticles), natural polymers (hyaluronic acid), and bio-adhesive molecule (dihydroxylphenylalanine, DOPA) to develop an intracellular ROS sensor for real-time imaging analysis." | |
| Lee is a post-doc researcher at the Bio-Inspired Nanomaterials Lab in the Department of Chemistry at Korea Advanced Institute of Science and Technology (KAIST). He is first author of a recent paper in Advanced Functional Materials (Fluorescent Gold Nanoprobe Sensitive to Intracellular Reactive Oxygen Species) that he wrote as a member of Tae Gwan Park's Nano-Biomaterials Lab at KAIST. | |
 |
|
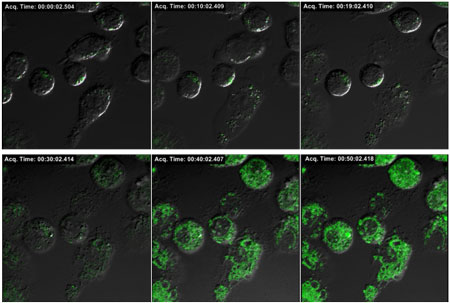
| Confocal microscope analysis of intracellular ROS generation in macrophage cells. Fluorescence recovery from intracellularly stable fluorescence gold nanoprobes upon lipopolysaccharide stimulation was monitored for two hours. Green fluorescence indicates the ROS induced cleavage of fluorescein-HA conjugates from the surface of gold nanoparticles. (Image: Dr. Hyukjin Lee, KAIST) | |
| Lee notes that the design of their intracellular ROS imaging probe was made possible by using biodegradable natural polysaccharide (hyaluronic acid) and mussel inspired bio-adhesive molecule (DOPA). | |
| " Since hyaluronic acid is highly sensitive and degradable by superoxide anions and hydroxyl radicals, it is readily cleavable by neighboring ROS and releases fluorescent dyes closely bound to the surface of gold nanoparticles, thus generating strong fluorescence recovery signals," he explains. "Meanwhile, DOPA assures strong adhesion of fluorescent labeled hyaluronic acid molecules onto the gold nanoparticles under intracellular reducing conditions, thereby minimizing the background fluorescence signals from ROS sensitive gold nanoparticles." | |
| In a live cell study, the Korean team has demonstrated real-time analysis of intracellular ROS generation in macrophages upon lipopolysaccharide (LPS) stimulation. "We observed LPS induced oxygen bursts in macrophages by confocal microscopic analysis and fluorometer measurement over a time scale of several hours," says Lee. | |
| As an alternative application of this novel nanoprobe, Lee and his collaborators used it to screen antioxidants with respect to their ROS-scavenging capacities. "We tested various antioxidants such as ascorbic acid, p-coumaric acid, quercetin, and a-tocopherol as model scavengers for ROS" says Lee. "By using chemically generated superoxide anion in a cell-free environment, we examined the possibility of high-throughput analysis of antioxidants using a mini-well plate with our gold nanoparticle probes." | |
| The researchers measured the fluorescence intensity from their nanoprobes in individual mini-wells with an assay volume of 10µl with a fluorescence image analyzer. They were able to observe enhanced fluorescence signals with increasing ROS concentration. | |
| Overall, the gold nanoprobes were very stable in a highly reductive environment and could be exploited for sensitive detection of chemically generated ROS as well as intracellular ROS generation in live cells. These intracellularly stable fluorescence gold nanoprobes are potentially useful for optical imaging of ROS and screening of their inhibitors such as antioxidant and superoxide dismutase. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
