| Posted: Jul 17, 2009 | |
Nanofluidics meets nanoplasmonic sensing |
|
| (Nanowerk Spotlight) In physics, a plasmon is the quasiparticle resulting from the quantization of plasma oscillations just as photons and phonons are quantizations of light and sound waves, respectively. As the name indicates, surface plasmons are those plasmons that are confined to surfaces. Controlling these surface plasmons has become increasingly attractive for optical signal processing, surface enhanced spectroscopy and sensor nanotechnology. For instance, the role of surface plasmon resonance (SPR) on resonant transmission through nanohole arrays has motivated their application as surface-based biosensors. | |
| "As compared to common SPR sensing, nanohole arrays present many advantages, including a smaller foot-print, lower limits of detection, denser integration, multiplexing, and collinear optical detection," David Sinton, an associate professor in the Department of Mechanical Engineering at the University of Victoria in Canada, explains to Nanowerk. "Arrays of nanoholes in a metal film exhibit unique optical properties in part due to the presence of surface plasmons – light waves confined near the metallic surface. Due to the confinement of the light near the surface, nanohole arrays have been employed to sense the binding of biological molecules, with application to biosensing." | |
| As an example, Sinton says that cancer screening may be accomplished using suitably coated nanohole array based sensors. One of the problems so far has been to transport an analyte of interest to a surface based sensor, and thus achieve a low limit of detection. | |
| New work by a team of scientists from Sinton's, Brolo's and Gordon's group at the University of Victoria, and the British Columbia Cancer Agency, has combined nanofluidics and nanoplasmonics for SPR sensing using flow-through nanohole arrays. This new format enables rapid transport of reactants to the active sensing surface and the array serves as a sieve. That is, the flow-through array efficiently collects and detects biomarkers from a very small volume of fluid. | |
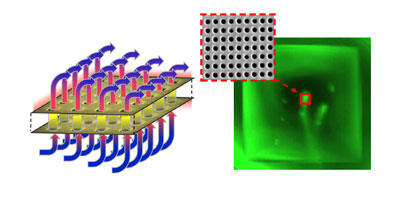 |
|
| Schematic of the flow-through nanohole arrays and fluorescence image showing a dye buffer solution streaming from the array. (Image: Dr. Sinton, University of Victoria) | |
| Sinton points out that their findings indicate a 6-fold improvement in response time as compared to the established method. | |
| The team has reported their findings in a recent paper in Analytical Chemistry ("Nanoholes As Nanochannels: Flow-through Plasmonic Sensing"). | |
| "Research to date has focused on arrays of dead-ended holes, or pits" says Sinton. "In terms of transport, previous nanohole array sensors didn't take advantage of their nanostructure. Our findings indicate that flow-through nanohole array format offers significant advantages with respect to the transport and sieving of biomarkers." | |
| The researchers fabricated their circular nanohole arrays by focused-ion beam milling through a 100 nm thick gold film thermally evaporated on free-standing 100 nm silicon nitride with a 5 nm thick chromium adhesion layer. By varying the milling parameters they were able to determine the conditions for fabricating arrays that allowed through-hole transport. | |
| This work has been motivated by the need for an effective cancer screening method where quantitative detection of multiple biomarkers is required in order to provide a diagnosis. Particularly motivating are such diseases that are treatable if detected early, yet generally asymptomatic until late stages. An example is ovarian cancer as studied by collaborators at the BC Cancer Trev & Joyce Deeley Research Centre. | |
| Sinton explains how the new device's combination of nanofluidics and nanoplasmonics works: "As fluidic elements, the nanohole arrays serve to parallelize the resistance and thus fluid handling and control are compatible between the parallel nanochannels and established microfluidic protocols. As optical elements, the nanohole arrays serve to detect the adsorption of a monolayer as well as the step-by-step multilayer assembly of biomolecules as required for biosensing applications." | |
| Sinton suggests that future work will focus on two areas. "Firstly, flow-through nanohole array based sensors have the potential for dense integration in a lab-on-chip format, but the implementation of that is not straightforward and is a current focus. Secondly, the transition from microscale to nanoscale channels that is involved with this new flow-through format offers some interesting physics. There are several potential opportunities to exploit these fundamental phenomena to achieve fluid pumping, analyte concentration and control of particles in the vicinity of the sensing surface with implications for sensing." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
