| Posted: Nov 24, 2010 | |
Step-by-step chemical transformations with logic gate nanoparticles |
|
| (Nanowerk Spotlight) Many nanostructures show to be responsive to external stimuli such as temperature, pH, or solvent polarity. An exciting field for nanotechnology researchers is the challenge of extending the scope of nanostructures with stimulus-responsive properties towards two main goals: 1) the fabrication of 'smart' nanoscale materials, for instance for self-healing surfaces or reversible structural transformation of self-assembled materials, and 2) the targeted and controlled delivery of therapeutics and diagnostics at the cellular level. | |
| Led by Adah Almutairi, a team from the Laboratory for Bioresponsive Materials at UC San Diego have now shown that two pH response moieties – a pH solubility switch and a pH labile group – can be incorporated into the backbone of polymers which can then be formulated into dual responsive nanoparticles encapsulating small hydrophobic molecules and larger protein payloads. | |
| Reporting their findings in a recent issue of ACS Nano ("Multiresponse Strategies To Modulate Burst Degradation and Release from Nanoparticles"), the researchers describe the dual pH response moieties function of their nanoparticles – to fine-tune their payload release and subsequent degradation – like a logic gate. | |
| According to Almutairi, the team hypothesized that developing a system that has two or more pH response mechanisms to a single triggering event would more finely tune the response to pH stimuli. | |
| Accordingly, they developed a dual pH responsive random co-polymer that utilizes two distinct mechanisms: the ability to switch from hydrophobic to hydrophilic (like poly-β-amino esters) and the triggering of acid–base-catalyzed hydrolyses (utilizing a ketal moiety). | |
| As nanoparticles they function akin to an AND logic gate. The β-aminoester backbone moiety provides a pH triggered solubility switch, only when this switch is "ON" does the ketal moiety also turn "ON" to undergo rapid acid catalyzed hydrolysis. | |
 |
|
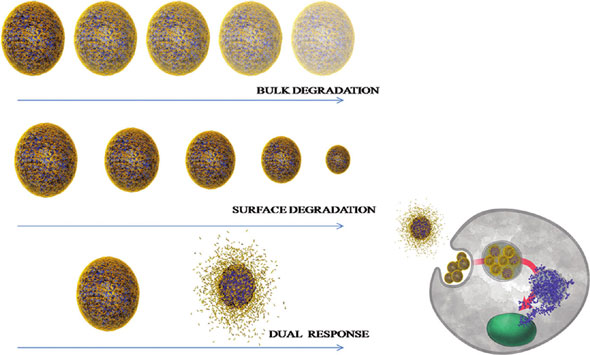
| Different release profiles from nanoparticles result in different cellular response. (Reprinted with permission from American Chemical Society) | |
| The researchers formulated nanoparticles from poly-β-aminoester ketal-2 which become hydrophilic at mildly acidic pH 6.5–5.0 and in turn lead to accelerated hydrolysis of the ketal moieties. In response to a single triggering event of a decrease in pH, the amine backbone undergoes a sharp hydrophobic–hydrophilic switch. This leads to an increase in uptake of water (bulk dissolution) and hence an increase in ketal hydrolysis (surface degradation). | |
| "The pronounced effect of a hydrophilic–hydrophobic balance is evidenced by the fact that our degradation times are significantly faster than that obtained for other hydrophobic polyketals," says Almutairi. "Furthermore, our dual pH response design showed better stability at physiological pH (7.4) than other hydrophilic polyketals, while maintaining the desired rapid degradation at acidic pH." | |
| The nanoparticle formulations were stable for 24 hours in healthy physiological pH, and upon reducing the pH to endosomal levels, pH 5, these dual responsive nanoparticles underwent a rapid and dramatic degradation followed by concomitant release of their payloads. | |
| Almutairi concludes that this system seems to be a promising vehicle for the administration of hydrophilic and hydrophobic payloads into target areas of the human body. | |
| "Continued efforts in designing optimal bioresponsive polymers would improve limitations in drug, diagnostic, and biopharmaceutical delivery. We are currently applying these novel systems to sense and image early stages of metabolic diseases and inflammation." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
