| Posted: Jan 03, 2011 | |
A nanoscale biofuel cell for self-powered nanotechnology devices |
|
| (Nanowerk Spotlight) Nanotechnology researchers working on self-powered nanodevices – nanoscale systems that scavenge energy from their surrounding environment – have been experimenting with various power sources ranging from piezoelectric systems ("Electricity-generating silicone implants could power electronic devices") to sound ("Nanotechnology energy generation using sound"). | |
| However, the most abundant energy available in biosystems is chemical and biochemical energy, such as glucose. Researchers in China have now reported a nanowire-based biofuel cell (NBFC) based on a single proton conductive polymer nanowire for converting chemical energy from biofluids into electricity, using glucose oxidase and laccase as catalyst. | |
| "We have demonstrated an innovative single nanowire biofuel cell for harvesting chemical/biochemical energy for powering in vivo nanodevices," Caofeng Pan tells Nanowerk. "The output of our NBFC is sufficient to drive pH, glucose or photon sensors. The high output power, low cost and easy fabrication process, large-scale manufacturability, high 'on-chip' integrability and stability demonstrates its great potential for in vivo biosensing." | |
| Pan is a post-doc researcher in Jing Zhu's Group at the Beijing National Center for Electron Microscopy and the Department of Materials Science and Engineering at Tsinghua University. He is first author of a recent paper in Advanced Materials ("Generating Electricity from Biofluid with a Nanowire-Based Biofuel Cell for Self-Powered Nanodevices") where the team, in collaboration with Georgia Tech's Zhong Lin Wang, introduces their nano biofuel cell. | |
| "In previous work on nanowire-based bio fuel cells, we have reported a platinum-catalyzed fuel cell ("Nanowire-Based High-Performance 'Micro Fuel Cells': One Nanowire, One Fuel Cell") and based on an individual nanowire" says Pan. "For our latest work, the whole cell can be accomplished at nano/micron scale, and the biofuel cell of a single nanowire generates an output power as high as 0.5-3 µW in glucose solution, in human blood and the juice of a watermelon. It has been integrated with a set of nanowire based sensors for performing self-powered sensing." | |
 |
|
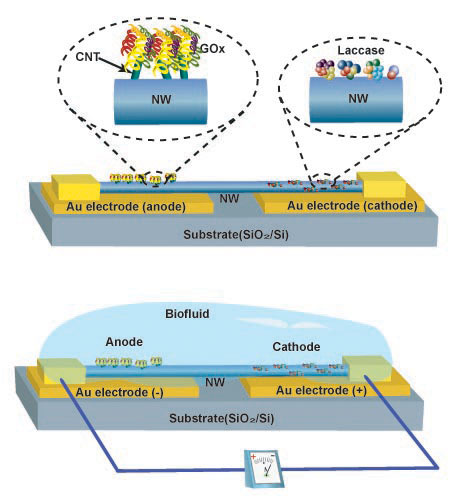
| Design of a single Nafion/poly(vinyl pyrrolidone) compound nanowire-based biofuel cell. a) The nanowire lies on a substrate (of any kind), with both ends tightly bonded to the substrate and outlet interconnects. GOx and laccase are used as catalysts in the anode and cathode region, respectively. b) The NBFC is immersed into a biofuel solution, two chemical reactions occur in the anode and cathode regions, creating a corresponding chemical potential drop along the nanowire, which drives the flow of protons in the nanowire and electrons through the external load. (Reprinted with permission from Wiley-VCH Verlag) | |
| The proton exchange membrane is the key component in many of today's fuel cells – and it is also a big obstacle to shrink the size of a fuel cell. | |
| Pan explains that, in the team's early fuel cell research, they fabricated Nafion nanowires, which have an enhanced performance of proton conductivity compared with Nafion film. The Nafion nanowires have a diameter about 100 nm to 1 µm, and a length of about 20 µm. However, it turns out to be very difficult to build a platinum-catalyzed fuel cell on such a small nanowire as the anode and cathode reacting area needs to be strictly separated. | |
| In subsequent work, the scientists introduced a new method – electrospinning – to fabricate Nafion nanowires. Electrospinning is a very powerful method to produce polymer nanowires (such as Nafion). | |
| "With this method, we can easily produce Nafion nanowires as long as centimeters" says Pan "Now it became very easy for us to build a fuel cell based on an individual nanowire." | |
| An additional approach to solve the miniaturization problem is to fabricate a biofuel cell instead of a fuel cell. In this case, an enzyme pair (such as GOx and Laccase) is used as catalysts. | |
| "The anode and cathode area need not to be separated again since enzymes are very selective to the reactants, for example, the GOx will only oxidize glucose, while Laccase will only reduce oxygen in the biofluid," Pan explains. "As a result, the size of the fuel cell can be reduced again." | |
| Studying the performance of the NBFC when directly interfacing with available 'biofuels' in the human body is necessary for exploring its potential applications in powering in vivo wireless nanodevices. The team found that their NBFC is strong enough to power nanowatts-consuming nanodevices, with the power output in glucose-containing PBS buffer solution reaching about 2.7 µW. The corresponding power output density is around 30 µW cm-2. The performance of the NBFC driven by blood glucose was 0.5 µW. The researchers attributed this decrease in blood compared to glucose/PBS solution to partial enzyme inhibition by several compounds presented in human blood. | |
| Additional studies showed that the NBFC works even using watermelon juice as the biofuel, with the output of the NBFC being similar to those obtained using glucose solution and human blood as biofuels. | |
| The team also showed that their nano biofuel cell can be directly integrated with a single nanowire-based nanosensor for building a self-powered chemical- or bio-sensor. By integrating an NBFC with a single nanowire-based pH sensor or glucose sensor fabricated using ZnO nanowire, they formed a self-powered nanosensor. | |
| These results show that the NBFC provides a new approach for self-powered nanotechnology that harvests electricity from the environment for applications such as implantable biomedical devices, wireless sensors, and even portable electronics that are important for biological sciences, environmental monitoring, defense technology and even personal electronics. | |
| Pan says that the team will now try to build a self-powered nanosystem that can be used in biological sciences, environmental monitoring, defense technology and even personal electronics. Such a self-powered nanosystem should include the nanodevices, power harvesting unit, electrical measurement system, data processing logic system, and possibly wireless communication unit (RF technology). Another objective they try to achieve is to improve the output of the biofuel cell to a level that allows it to power a heart cardiac pacemaker by generating power from human blood. | |
| Reference: Pan, C., Wu, H., Wang, C., Wang, B., Zhang, L., Cheng, Z., Hu, P., Pan, W., Zhou, Z., Yang, X., & Zhu, J. (2008). Nanowire-Based High-Performance "Micro Fuel Cells": One Nanowire, One Fuel Cell Advanced Materials, 20 (9), 1644-1648 DOI: 10.1002/adma.200700515 | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
