| Posted: Aug 01, 2013 | |
A sustainable nanotechnology design for micro-sized microbial fuel cells |
|
| (Nanowerk Spotlight) Microbial fuel cells are a prime example of environmental biotechnology that turns the treatment of organic wastes into a source of electricity. Fuel cell technology, despite its recent popularity as a possible solution for a fossil-fuel free future, is actually quite old – the principle was discovered in 1838 and the first fuel cell was developed in 1843. The operating principle of a fuel cell is fairly straightforward: it is an electrochemical energy conversion device that converts the chemical energy from fuel (on the anode side) and oxidant (on the cathode side) directly into electricity. | |
| Today, there are many competing types of fuel cells, depending on what kind of fuel and oxidant they use. For instance, a hydrogen fuel cell uses hydrogen as fuel and oxygen as oxidant. In microbial fuel cells (MFCs), the naturally occurring decomposing pathways of electrogenic bacteria are used to both clean water and produce electricity by oxidizing biological compounds from wastewater and other liquid wastes, even urine. | |
| "Micro-sized microbial fuel cells are essentially miniature energy harvesters requiring only the insertion of a liquid feed source containing organic materials for the bacteria to feed," Muhammad Mustafa Hussain, an Associate Professor of Electrical Engineering at King Abdullah University of Science and Technology (KAUST) in Saudi Arabia, explains to Nanowerk. "As a new technology, a full range of microbial fuel cell conditions and materials must be rapidly tested to determine the optimal parameters for maximum power production and future commercialization. From that perspective, micro-sized MFCs offer a unique miniature platform for rapid testing of MFC components." | |
| In new work, Hussain and his team have now demonstrated a sustainable and practical design for a micro-sized microbial fuel cell. Reporting their work in the July 30, 2013 online edition of ACS Nano ("Sustainable Design of High Performance Micro-sized Microbial Fuel Cell with Carbon Nanotube Anode and Air Cathode"), the team has successfully integrated multi-walled carbon nanotubes (MWCNT) into the anode of a completely mobile micro-sized MFC using an air cathode. | |
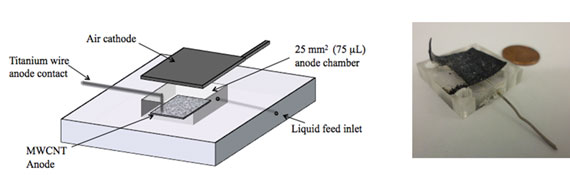 |
|
| (a) Schematic of the 75 µL micro-sized microbial fuel cell with MWCNT on silicon chip anode and air cathode. Gold and Nickel on silicon chip anodes were also tested and compared in the same set up; (b) Photograph of MWCNT on silicon chip microbial fuel cell in plastic encasing with titanium wire contact visible as well as the black air cathode compared to a US penny. (Reprinted with permission from American Chemical Society) | |
| With a focus on sustainability and low-cost usability, the researchers designed their MFC as a one-chamber device by removing the proton exchange membrane and replacing the cathode/chemical electron acceptor combination with an air cathode and ambient oxygen electron acceptor, thus making the entire device small enough for system-on-chip functionality. | |
| "We have used oxygen instead of hazardous chemical ferricyanide, which eliminates otherwise required continuous flow of liquid chemicals," says Justine Mink, a PhD student in Hussain's Integrated Nanotechnology Laboratory, and the paper's first author. "This is the first time oxygen has been used in micro-sized MFC. By comparing the same air cathode set up with the most commonly used but expensive gold anode as well as an inexpensive metal nickel anode we were able to confirm that air cathodes in microsized MFCs are feasible even without a membrane and that the devices are durable and long-lasting." | |
| She points out that, during their experiments where they tested their MFCs for over 15 days, the used MWCNT anode outperformed the others in current and power production most importantly due to its increased surface area. | |
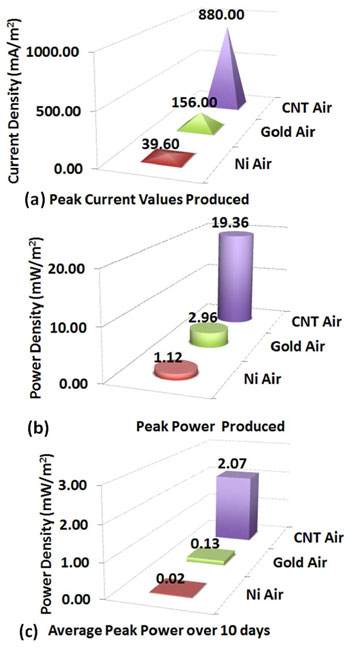 |
|
| Maximum current densities produced by the devices (a) are about 800% higher for the MWCNT anode compared to the gold and more than 2200% higher compared to the nickel anode. Maximum power densities (4b) indicate that the MWCNT anode produced more than 600% the power of the gold anode and 1900% the power of the nickel anode. Their peak power values over a 10 day period (4c) show that all devices were able to have reproducible and stable power but not at the values of their peak power achieved indicating further need for optimization within the micro-size MFC. (Reprinted with permission from American Chemical Society) | |
| Having system-on-chip functionality on their mind, Hussain's group used CMOS compatible processes to fabricate their anodes with pure carbon nanotubes on silicon. The material selection for the cathode, like that of the anode, requires a highly conductive material and is most typically carbon. In this case, the team used a specially designed and nanoengineered carbon cloth air cathode. This is the first time an air cathode has been integrated directly onto a silicon based MFC chip. | |
| Applications of this new microbial fuel cell design will be found in two areas: as rapid testing tools for MFC components for macroscale designs; and as onboard power source for lab-on-chip and point-of-care diagnostic devices. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
