| Posted: Jun 15, 2015 | |
3D-printed programmable release capsules |
|
| (Nanowerk Spotlight) As we are seeing an increasing number of 3D-printing appearing in nanotechnology applications (see for instance these recent Nanowerk Spotlights: 3D-printed graphene for electronic and biomedical applications; 3D-printed 'smart glue' leverages DNA assembly at the macroscale; or Fully 3D-printed quantum dot LEDs), researchers are expanding this fabrication technique to more and more areas. | |
| A recent example is the introduction of a novel 3D-printing based method to produce highly monodisperse core/shell capsules that can be loaded with biomolecules such as therapeutic drugs. | |
| "Our method provides us with robust control over particle properties, passive release kinetics, and particle distributions throughout a 3D matrix," Michael McAlpine, an associate professor in mechanical engineering at the University of Minnesota, tells Nanowerk. "Furthermore, we render these capsules stimuli-responsive by incorporating gold nanorods into the polymer shell, allowing for highly selective photothermal rupture and triggered temporal release of the biomolecular payload." | |
| McAlpine and his group, working with researchers from Washington University in St. Louis, published their findings in the June 4, 2015 online edition of Nano Letters ("3D Printed Programmable Release Capsules"), where they describe the use of 3D printing to hierarchically order polymers, biomolecules, and nanomaterials into hybrid functional materials in a scalable manner. | |
 |
|
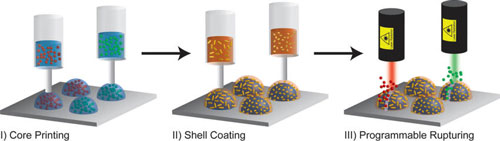
| Programmable printing and rupturing of capsules: (I) multiplexed arrays of aqueous cores containing biomolecular payloads are printed directly on a solid substrate; (II) PLGA solutions containing AuNRs of varying lengths are dispensed directly on the aqueous cores, forming a solid stimuli-responsive shell; (III) the capsules are selectively ruptured via irradiation with a laser wavelength corresponding to the absorption peak of the nanorods. (Reprinted with permission by American Chemical Society) (click on image to enlarge) | |
| "Our technique is enabled via the 3D-printing of multiplexed arrays of biomolecule-loaded capsules, along with tunable and orthogonal laser-triggered rupture and release of active biomolecules," Maneesh K. Gupta, a former member of McAlpine's group and now a research scientist at Air Force Research Laboratories, explains. "One can imagine filling the capsules with molecules such as drugs, nucleic acids, enzymes, growth factors, cell markers and other functional proteins, into a hydrogel ambient." | |
| Adds Fanben Meng, a postdoc researcher in McAlpines Lab and co-first author with Gupta: "The advantages of our 3D printing-based method include: 1) highly monodisperse capsules; 2) efficient encapsulation of biomolecular payloads; 3) precise spatial patterning of capsule arrays: 4) 'on the fly' programmable reconfiguration of gradients; and 5) versatility for incorporation in hierarchical architectures." | |
| An interesting feature of these capsules is the control of the spatial and temporal release of payloads. This is achieved by incorporating gold nanorods in the shell. Using specific wavelengths determined by the length of the nanorods, the rods can be heated which subsequently ruptures the shell. | |
| The 3D printing technique itself also provides precise control over the capsule volumes and architectures. | |
| "This work provides a promising solution to generating multiplexed spatiotemporal molecular gradients in 3D architectures, which is significant to mimic the dynamic microenvironment surrounding cells in natural tissues, as living organisms guide tissue development through highly orchestrated gradients of biomolecules that direct cell growth, migration, and differentiation in 3D matrices," notes Meng. | |
 |
|
| 3D printing of hierarchically multiplexed capsule arrays. Top row: Schematic illustrating an emulsion ink-based method to 3D print complex capsule arrays. The emulsion ink is prepared by directly dispersing the aqueous core in the PLGA solution. The hydrogel and emulsion inks are sequentially printed in a layer-by-layer manner to form a 3D structure. Bottom row: Optical images of 3D multiplexed capsule arrays directly printed in cylindrical and square hydrogel matrices, respectively (colors of the capsules are from food dyes in the dispersed cores). (Image: University of Minnesota) | |
| The researchers expect that this platform of 3D printed programmable release capsules will be useful in applications such as dynamic tissue engineering, 3D printed drug delivery systems, synthetic/artificial tissues, programmable matter, and bionic nanosystems. | |
| Beyond this, another important application area could be combinatorial screening of biomolecular gradients – drugs, toxins, pollutants, etc. – against cell types. | |
| A particularly far-reaching example would be to imagine having a collection of stem cells, which could be triggered using a red laser into a heart, or with a green laser into a liver. | |
| "Our work was motivated by the fact that living systems utilize exquisite control of biomolecular gradients to control cell fate and ultimately enable complex functional tissues," says McAlpine. "We believe that replicating such control is a key to many future advances in bioengineering." | |
| "There has been tremendous prior work on utilizing microfluidics, particle encapsulation, and stimuli-responsive materials to address some of these challenges," he adds. "Our findings offer a novel perspective by offering a 3D printing based approach to solve these challenges, which has advantages in precision control over volumes, spatial distributions, and diversity of materials including nanomaterials and biomolecules." | |
| A next stage of the investigation now requires studies regarding the biocompatibility and feasibility of using 3D printed capsules to control cell fate, from the individual cell level to the level of tissue engineering. | |
| As the team points out, beyond biological studies, there is a tremendous amount of work to be done to enhance the material and functional properties of the capsules. For example, improving the resolution and spatial alignment of these printed capsules, developing stimuli responsive shells with reversible payload release properties, and developing other approaches to stimulate the capsules, such as using electrical signals rather than light triggering. | |
| This work was funded by the NIH, AFOSR, and Intelligence Community. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
