| Posted: Jan 23, 2017 | |
A nanotechnology approach to purifying liquid crystals |
|
| (Nanowerk Spotlight) Liquid crystals used in modern devices such as laptops, tablets and smartphones typically contain a small fraction of ionic contaminants. These ion contaminants can originate from multiple sources during the chemical synthesis of materials, in the process of assembling the device, and in its daily use. | |
| In the case of liquid crystal displays (LCDs), mobile ions in liquid crystals lead to such undesirable effects as image sticking, image flickering, and slow response. | |
| That is why ions in liquid crystals are not desirable and their concentration should be kept as low as possible. | |
| A promising solution to reduce the concentration of mobile ions in liquid crystal devices can be found by merging liquid crystals and nanotechnology. | |
| Nanomaterials dispersed in liquid crystals can trap and immobilize ions thus providing a permanent purification of liquid crystals from ions. This concept of the ion capturing effect in liquid crystals doped with nanoparticles was verified by numerous research groups around the globe (see this review for more details: Crystals, "Nano-Objects and Ions in Liquid Crystals: Ion Trapping Effect and Related Phenomena"). | |
| "An interesting and practically important question is what happens to the ion capturing effects if the temperature changes. Does the ion capturing efficiency of nanomaterials remain the same at low and high temperatures?" Yuriy Garbovskiy, PhD, a researcher at the UCCS BioFrontiers Center & Department of Physics, University of Colorado, tells Nanowerk. "This is very important and practical question. For example, on a sunny day, you decided to take your lap top, go to the park and work on your project there. Will ions in liquid crystals and their sensitivity to temperature ruin your day?" | |
| A recently published paper in Liquid Crystals ("Nanoparticle enabled thermal control of ions in liquid crystals") provides an answer to some of these questions (unfortunately, you and your laptop in the park were not discussed) by modeling the temperature dependence of the concentration of mobile ions in liquid crystals doped with nanoparticles. | |
| The basic idea of this work is the use of the Langmuir adsorption isotherm in conjunction with the Arrhenius equation, and the quantification of the contamination of nanoparticles by means of the dimensionless contamination factor. | |
| An interesting aspect of this paper is the consideration of both 100% pure and contaminated with ions nanoparticles. | |
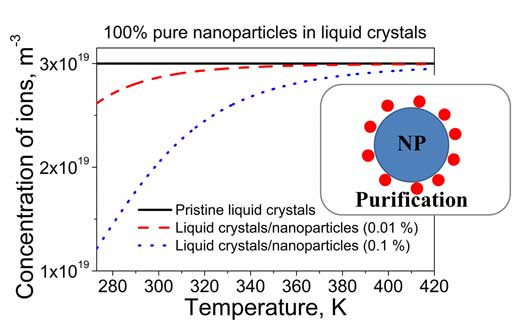
| Garbovskiy explains: "Typically, ionic species in thermotropic liquid crystals are nearly fully ionized. As a result, the concentration of mobile ions practically does not depend on the temperature (Figure 1 solid curve). Interestingly, the same liquid crystals doped with nanoparticles exhibit totally different behavior. The concentration of mobile ions exhibits strong temperature dependence (Figure 1, dashed and dotted curves)." | |
 |
|
| Figure 1. The temperature dependence of the concentration of mobile ions in pristine liquid crystals (solid curve), and liquid crystals doped with 100% pure nanoparticles (dashed and dotted curves). (Image: Dr. Yuriy Garbovskiy, University of Colorado, Colorado Springs) | |
| He points out that a very important aspect is that 100% pure nanoparticles dispersed in liquid crystals lower the concentration of mobile ions in liquid crystals (Figure 1, dashed and dotted curves are below the solid curve). This ion capturing effect results in the permanent purification of liquid crystals from ions. However, this effect depends on the temperature as is evidenced by Figure 1, dashed and dotted curves. | |
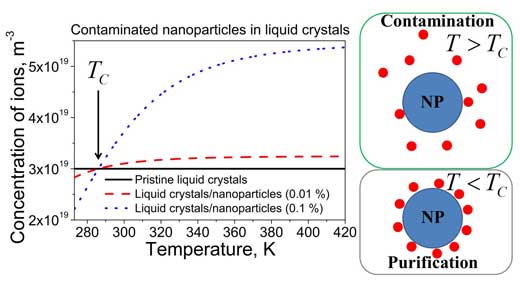
| The use of nanoparticles contaminated with ions prior to their dispersion in liquid crystals results in more complicated behavior shown in Figure 2. | |
| In this case, depending on the temperature, both ion trapping (the purification of liquid crystal from ions) and ion releasing regimes (the contamination of liquid crystals with ions) are possible. | |
| The purification regime is observed if the temperature of liquid crystals T is below the temperature Tc. The contamination regime holds true above this temperature (T > Tc). | |
 |
|
| Figure 2. The temperature dependence of the concentration of mobile ions in pristine liquid crystals (solid curve), and liquid crystals doped with contaminated pure nanoparticles (dashed and dotted curves). (Image: Dr. Yuriy Garbovskiy, University of Colorado, Colorado Springs) | |
| "The key finding here is that, while the dispersion of 100% pure nanoparticles in liquid crystals leads to the temperature dependent purification only (Figure 1), the use of contaminated nanoparticles results in the temperature-driven switching between the purification and contamination regimes enabling thermal control of ions (Figure 2)," Garbovskiy says. | |
| He adds that, regardless the ionic purity of nanodopants, the concentration of mobile ions in liquid crystal nanocolloids increases towards the saturation level as their temperature goes up. | |
| In the case of 100% pure nanoparticles, the magnitude of this saturation level equals the concentration of ions in pristine liquid crystals (Figure 1, dashed and dotted curves approach solid curve at higher temperatures). The temperature induced release of ions by contaminated nanoparticles in liquid crystals increases the above-mentioned saturation level. In this case the magnitude of the saturation level is proportional to the concentration of contaminated nanoparticles in liquid crystals (Figure 2, dashed and dotted curves). | |
| "These results add to our understanding of physics of liquid crystals doped with nanoparticles," notes Garbovskiy. "An important practical implication is the dependence of the ion trapping efficiency of nanomaterials on the temperature. This dependence should be considered by R&D engineers using liquid crystal / nanoparticle colloids in their devices." | |
| For example, the operating temperature of the device should be set below the temperature Tc to ensure nanoparticles act as ion capturing agents. This fact is also important for the design of electro-optical devices utilizing nanoparticles and liquid crystals. | |
| It should be noted that ions in liquid crystals are not always a bad thing. For example, some devices such as optical switches utilizing the effects of light scattering rely on ions in liquid crystals. This type of applications could benefit from the use of contaminated nanoparticles and temperatures T > Tc. | |
| Thus, nanoparticle enabled thermal control of ions in liquid crystals can be used for a variety of display and non-display applications of liquid crystals. | |
| "We obtained the results – temperature induced increase in the concentration of mobile ions in liquid crystals doped with nanoparticles – assuming certain conditions naturally expected for the ion trapping nanomaterials," says Garbovskiy. "These conditions can be expressed by the positive value of the parameter ΔE defined as a difference between desorption activation energy and adsorption activation energy (for example, in the cases shown in Figures 1 and 2 this parameter equals 0.3 eV)." | |
| "However" he adds, we can also expect an opposite case characterized by the negative value of this parameter." | |
| This scenario was analyzed in another paper in Liquid Crystals ("Ions in liquid crystals doped with nanoparticles: Conventional and counter intuitive temperature effects"). | |
| According to this work, liquid crystals doped with nanoparticles characterized by the negative value of the parameter ΔE exhibit a temperature induced decrease in the concentration of mobile ions. | |
| This paper also provides a guidance on the selection of suitable nanomaterials exhibiting the scenario ΔE < 0. | |
| "Research on ions and nano-objects in liquid crystals is very diverse; it can go different ways by exploring various nanoparticles and liquid crystals," concludes Garbovskiy. "Practically important is an exploration of nanoparticles in high resistivity liquid crystals. In our work, we suggest the use of polymer nanoparticles as possible candidates to observe the temperature induced decrease in the concentration of mobile ions in liquid crystal nanocolloids. It could be very interesting to test this prediction experimentally." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
