| Posted: May 24, 2017 | |
Electrochemical energy storage with porous one-dimensional (1D) nanomaterials |
|
| (Nanowerk Spotlight) The development of porous one-dimensional (1D) nanomaterials (check our nanotechnology FAQ to learn more about number of dimensions of a nanomaterial) with designed properties and architectures has led to significant advances in electrochemical energy storage. | |
| In a recent review article in Advanced Materials ("Porous One-Dimensional Nanomaterials: Design, Fabrication and Applications in Electrochemical Energy Storage"), the authors highlight the state-of-the-art on porous 1D nanostructures, from methodologies for rational and controllable synthesis (electrospinning, liquid phase method, template-assisted method, chemical deposition method, and chemical etching) to their successful application in different types of energy storage devices. | |
| They suggest that porous 1D nanostructures will overcome the limitations of many electrode materials, so that high capacity, fast charge and discharge, and long cycle life can be realized. | |
 |
|
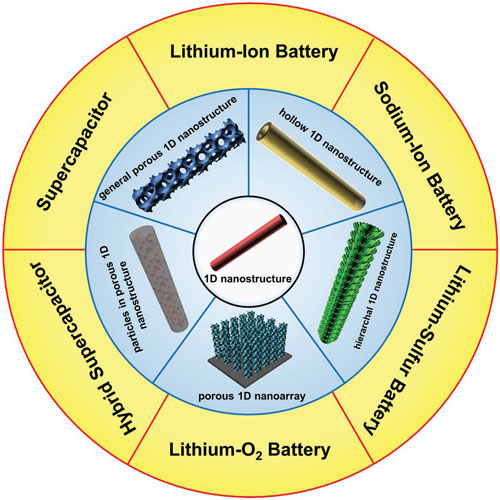
| Porous 1D nanostructures and their applications in electrochemical energy storage. (© Wiley-VCH Verlag) | |
Alkali ion batteries |
|
| In alkali ion batteries (LIBs and SIBs), porous 1D nanostructures with much shorter bi-continuous ion and electron transport pathways benefit high rate applications. Concurrently, the development of a robust porous structure offers the advantages of accommodating large volume changes which prevent structure collapse and improve cycle life. | |
| Outstanding electrochemical performance has been reported using a variety of different porous 1D nanomaterials as they can overcome certain limitations: (1) the poor ionic and electronic conductivity of electrode materials; (2) the interfacial impedance between electrode and electrolyte which arise from SEI layers forming on the interface of active materials; and (3) low volumetric energy density. | |
| The rational design of porosity and architecture leads to materials with properties that enable fast ion diffusion and rapid electron transport, reduce the exposure of active materials to the electrolyte and use assembly methods to increase volumetric energy density. | |
Advanced lithium batteries |
|
| Advanced lithium batteries (Li-S and Li-O2 systems) that offer higher energy density than LIBs can also benefit from using porous 1D nanostructures. Through nanocasting, sulfur can be embedded in porous nanofibers, establish strong adsorption properties and thereby inhibit the “shuttle effect” in polysulfide systems. | |
| A gradient macro/meso/micro-porous structure with increased sulfur loading is able to significantly enhance the energy density of Li-S batteries. In Li-O2 batteries, porous nanowires provide large surface area, continuous O2 and electrolyte diffusion channels, as well as good electron transport. | |
| Nanowires also provide numerous deposition/decomposition sites for reaction products enabling improved electrochemical performance. | |
Supercapacitors |
|
| The electrochemical performance of supercapacitors that store charge by double layer or redox processes has been improved by using porous 1D nanostructures. | |
|
Carbon-based EDLC materials with heteroatom-doping are able to create more redox sites to increase the energy density. In addition, well-constructed porous nanowires enable intrinsic and extrinsic pseudocapacitor materials to achieve improved performance by implementing short diffusion paths, large ion adsorption and fast electrolyte access to redox-active sites. Constructing electrodes with porous 1D nanostructures that enable bi-continuous transport of electrons and ions will be beneficial for fabricating high energy density hybrid supercapacitors whose cathodes and anodes effectively combine the advantages of pseudocapacitive materials, especially for intercalation pseudocapacitance. "More systematic studies, both theoretical and experimental, are needed to optimize the design of these nanostructures for specific applications," the authors conclude. "For large scale and low cost production, the required synthesis procedures, which are relatively complex, must be simplified and engineered. We believe that high-performance energy storage devices based on advanced porous 1D nanomaterials will be an important direction for the future." |
|
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
