| Posted: Jul 26, 2017 | |
Nanotechnology for neural interfaces |
|
| (Nanowerk Spotlight) Neural interfaces establish direct communication between the central nervous system (CNS) and a sovereign, man-made digital system. This technology is perhaps the most important advance in the study and treatment of the brain is the development of the neural interface. | |
| Different biomedical devices implanted in the central nervous system, so-called neural interfaces, already have been developed to control motor disorders or to translate willful brain processes into specific actions by the control of external devices (read more: "Nanotechnology coming to a brain near you"). | |
| These interfaces promote a neurophysiological understanding and provide a clinical means for detecting or treating neurological symptoms and diseases. They could help increase the independence of people with disabilities by allowing them to control various devices with their thoughts – not surprisingly, the other candidate for early adoption of this technology is the military. | |
| Neural interfaces, or brain-machine interfaces (BMIs), can preserve the function of impaired neuronal tissues by translating nervous system signals into quantities that can be computationally understood. | |
 |
|
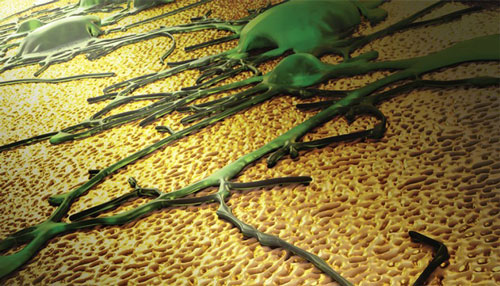
| The image depicts a neuronal network growing on a nanotextured gold electrode coating. The topographical cues presented by the coating preferentially favor spreading of neurons as opposed to scar tissue. This feature has the potential to enhance the performance of neural interfaces. (Image: Ryan Chen/LLNL) (read more: Implantable neuronal electrode nanocoating good as gold) | |
| Notwithstanding the concerns of use of nanotechnologies in the field of brain-machine interfaces, their comprehensive design principle is to augment or restore one or more of the three interrelated biological complications that arise from neural impairment: sensory malfunction, loss of motor control, or disease-elicited intellectual changes. | |
| The success of such a device relies on understanding the principles of bioelectrical transduction, neural modulation through computational calibration, and acclimating signal processing. | |
| The ideal neural electrodes should be designed to have a small volume to minimize damage to the tissue, while maintaining high sensitivity, i.e., high signal-to-noise ratios. However, it is nearly impossible to satisfy these competing requirements with conventional microfabrication methods and materials. | |
| Nanotechnology fabrication methods can overcome these limitations by producing electrodes with an extremely high surface to volume ratio, i.e., more probe units within the same volume, resulting in unprecedented specificity. | |
| A lot of research is already taking place in this field, for instance by revolutionizing brain-computer interfaces using silicon electronics. This work is part of DARPA's Neural Engineering System Design (NESD) program that aims to develop an implantable neural interface able to provide advanced signal resolution and data-transfer bandwidth between the brain and electronics. | |
| In a new review article, just published in Advanced Functional Materials ("Nanotechnology and Nanomaterials for Improving Neural Interfaces"), the authors provide an overview of materials recently developed – specifically nanomaterials – that vastly enhance electrical properties, mechanical properties, location specificity, cytocompatibilty and biocompatibility to advance our understanding of the central nervous system and enable greater control over diseased/damaged states compared to current technologies. | |
| After discussing the current status of the biomaterial neural tissue interface, the authors focus on micro- and nanofabrication methods for electrode fabrication and techniques for evaluating and characterizing these electrodes. | |
| A large section of the article is dedicated to an overview of nanomaterials used for neural electrodes: graphene, carbon nanotubes, and hybrid nanomaterials. | |
 |
|
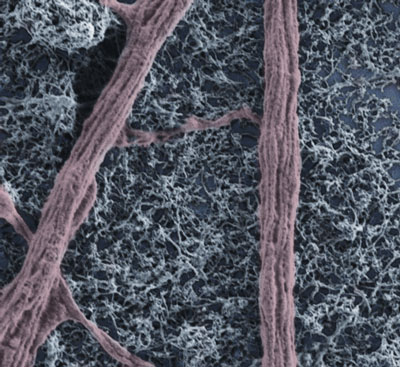
| Modified scanning electron microscopy image of spinal explant peripheral neuronal fiber on a multi-walled carbon nanotube substrate. (Image: Prato-Ballerini research group) (read more: Exploring the complexity of nanomaterial-neural interfaces) | |
| As the authors conclude their review, they point out that several strategies could prove promising for the development of improved neural interfaces. First, the optimization of electrode geometrical properties, including size, volume, geometry, and surface morphology, have been shown to influence, in one way or another, biocompatibility and electrical properties of electrodes. These improvements could be achieved by adopting more advanced nanofabrication techniques and surface modification methods capable of producing electrodes deemed far less biologically invasive. | |
| Second, the development and employment of novel nanomaterials such as carbon-based materials, conducting polymers, hydrogels, and hybrid materials that possess superior physical properties and biocompatibility have the ability to overcome the limitations that conventional metallic and semimetal materials are facing, i.e., great mechanical mismatch and gliosis. | |
| Third, the delivery of pharmaceutical agents (such as anti-inflammatory drugs) and/or the incorporation of bioactive coatings that encourage active neuron ingrowth may reduce reactive tissue responses and promote implant-tissue integration. | |
| In addition, chief among these trends is the development of wireless electrodes that enable prolonged recording and stimulation activities in the absence of bulky wires and attachments. Such technology could significantly improve our understanding of the brain by allowing continuous monitoring of neural signals during activities in which conventional wired electrodes would be prohibitive. | |
| Wireless electrodes could also enable the continuous stimulation of damaged or diseased brains to possibly restore functions of the CNS to people in need. | |
 |
|
| Arrays of semiconductor tubes can guide neuron outgrowth (through the tube) and signal propagation (shown as lightnings) among them. (Image: Minrui Yu, University of Wisconsin-Madison) (read more: Tunnels for neurons to guide brain-electronics interface studies) | |
| Additionally, researchers are incorporating natural materials within newly developed micro- and nanoelectrodes to improve biocompatibility for prolonged applications. | |
| Experimental work is underway to address the challenges of low organic semiconductor carrier mobility, a shortage of data describing noise interference in organic transistors, and the lack of long-term in vivo studies characterizing the interfacial implant region. | |
| It is anticipated that advancements in these areas will lead to an improved ability to monitor, stimulate, and record neurophysiological signals. This in turn may enable a paradigm shift in the way that people with disorders of the CNS are diagnosed and treated. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
