| Posted: Sep 08, 2017 | |
A nanotechnology approach to augment nerve regeneration |
|
| (Nanowerk Spotlight) Microtubules are architectural struts in nerves. They have dynamic parts that constantly assemble and disassemble, but also stable parts that remain assembled. In recent years, researchers working in neurobiology have been intrigued by the idea of microtubule-stabilizing drugs as a therapy to augment nerve regeneration. | |
| "Previous authors proposed that damaged nerves could be made to regenerate by using drugs that make the microtubules in the nerve more stable," Peter Baas, a professor in the Department of Neurobiology & Anatomy at Drexel University College of Medicine, tells Nanowerk. "In our recent work, we show that a better idea is to increase the amount of the dynamic parts of the microtubules. We do this by reducing the levels of fidgetin, a protein that normally exists in nerves to keep the dynamic parts of microtubules from elongating too much." | |
| As Baas and his collaborators report in Scientific Reports ("Nanoparticle Delivery of Fidgetin siRNA as a Microtubule-based Therapy to Augment Nerve Regeneration"), to reduce fidgetin levels, they use a nanotechnology approach. | |
 |
|
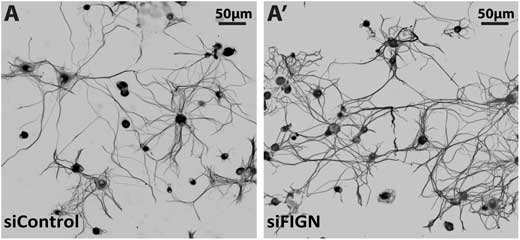
| Microtubule mass is increased in the axons of adult DRG neurons as a result of fidgetin knockdown. (A) Representative images of neuronal β-III-tubulin IF-staining show more extensive axonal outgrowth after fidgetin knockdown compared to control, and also show denser microtubule mass within axons, as indicated by more intense IF-staining. Shown are inverted images with black and white reversed for enhanced clarity. (© Nature Publishing Group) (click on image to enlarge) | |
| The two key elements in this approach are the novel idea of reducing fidgetin to increase dynamic parts of microtubules – as opposed to microtubule-stabilizing drugs – and the use of a siRNA-encapsulated nanoparticle delivery platform. | |
| The team uses RNA Interference, which stops new fidgetin from being synthesized, delivered via nanoparticles. The nanoparticles readily enter cells, tissues and organs in a manner that is potentially superior to other methods of delivery, such as viruses. | |
| "Based on a hydrogel/sugar glass composite, our delivery platform is a hybrid nanoparticle capable of encapsulating and controllably releasing a broad range of therapeutically relevant materials ranging from gaseous nitric oxide to larger macromolecules such as chemotherapeutic agents and phosphodiesterase inhibitors," Baas notes. | |
| He points out that the composition of the nanoparticles can be varied so that they release their load at different rates, which means that treatment regimens can be refined and adjusted accordingly. | |
| According to the team, fidgetin reduction can be used to augment nerve regeneration in a variety of injury situations, such as spinal cord injury, traumatic brain injury, and peripheral nerve injury. | |
| While the present paper only deals with a cell culture model (adult primary DRG neurons), the researchers are planning now to take the method into animal models. | |
| "The big challenge is not only to get the nerves to grow better but to enable them to find their appropriate target tissues to enable functional recovery," concludes Baas. "Given that the dynamic parts of microtubules are known to be very important for this, we think fidgetin reduction has a great chance of helping where nothing else has to date." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
