| Posted: Sep 22, 2017 | |
Designing next-generation neural interfaces with graphene |
|
| (Nanowerk Spotlight) In a previous Nanowerk Spotlight ("Nanotechnology for neural interfaces") we looked at the use of nanotechnologies in the field of brain-machine interfaces, their comprehensive design principle is to augment or restore one or more of the three interrelated biological complications that arise from neural impairment: sensory malfunction, loss of motor control, or disease-elicited intellectual changes. | |
| All currently used neural interface devices are designed to perform a single function: either record activity or electrically stimulate tissue. | |
| In order to fully exploit the potential of neural interfaces, the forthcoming generation of devices is expected to simultaneously offer multiple functionalities, including recording and stimulation of electrical activity, recognition of neurotransmitters, neuromodulators and other neurologically relevant biomolecules, as well as the capability for controlled drug delivery. | |
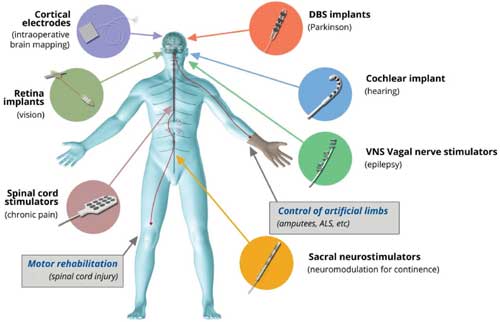 |
|
| Clinically developed neural interfaces. Schematics of the actual devices are shown (not to scale) along with each disease indication (in parentheses) mainly used or developed for. (© Wiley-VCH Verlag)(click on image to enlarge) | |
| Graphene and other two-dimensional (2D) materials possess an array of properties (flexibility, electrical mobility, large surface area available for interaction with the neuronal components and amenable to surface modifications) that can enable enhanced functional capabilities for neural interfaces. | |
| A paper in Advanced Materials ("Graphene in the Design and Engineering of Next-Generation Neural Interfaces") provides an overview of how graphene-based versatile platforms could help address many of the current challenges in neural interface design. | |
| Any neural interface designed for implantation should be as minimally invasive as possible, allow for a facile surgical procedure, and provide efficient and consistent activity for the duration of its functional lifetime. There are basically three major technological challenges required to achieve sufficient levels of efficacy: | |
|
|
|
| Because of their electrical and electrochemical performance and their suitability for integration into flexible devices, graphene-based materials constitute a versatile platform that could help address many of the above-mentioned challenges in neural interface design. | |
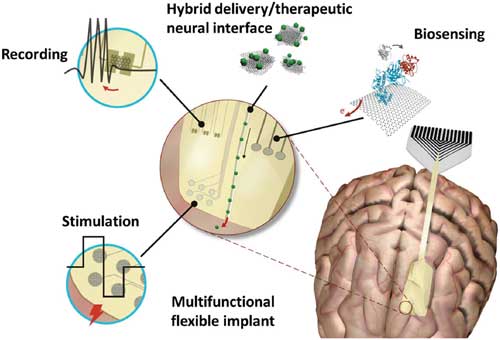 |
|
| Neural interface functionalities enabled by incorporation of graphene material. By utilizing the inherent properties that graphene materials offer, such as large surface area, flexibility, versatility for chemical functionalization and excellent performance of electronic devices, a variety of functionalities like recording, stimulation, biosensing, and drug delivery at the neural interface could be engineered in various combinations. (© Wiley-VCH Verlag)(click on image to enlarge) | |
Graphene-Based Materials for Neural Stimulation |
|
| Considering the capacitance and charge injection values of graphene, structured or 3D films of graphene-based materials, as well as composite films including graphene, show significant potential. | |
Graphene for Electrical Neural Recording |
|
| Graphene exhibits electrochemical capabilities for neural recording similar to platinum or gold, which have been for long the standard electrode materials for neural recording. In contrast to standard metals used in neural interfaces, graphene offers an unique advantage for recording applications: whereas metals only allow for designs based on the electrode configuration, graphene can be used to fabricate sensors based on a field-effect transistor (FET) configuration. | |
Graphene for Controlled Drug Delivery at the Neural Interface |
|
| Graphene and its derivatives are actively being explored in the field of therapeutic agent transport as nanoplatforms able to carry compounds of biological activity to specific cell populations and intracellularly. These and other strategies could be employed to engineer smart coatings for neural implants able to release biologically active molecules, to improve surface softness, and enhance neural recruitment and the overall biocompatibility of the implant. | |
Graphene for In Situ Biosensing |
|
| Due to its electronic properties and large surface to volume ratio, graphene has been extensively studied in the context of biosensing applications, and a broad variety of devices exhibiting high sensitivity, low noise, and low detection limits have been reported. The main challenge in most biosensing designs is tailoring specificity of the recognition event. This has to be achieved by a controlled chemical modification of graphene to enable the specific recognition of neurotransmitters, neuromodulators, or other biomarkers. | |
| The authors conclude that, given their versatility, which results from a remarkable combination of physico-chemical, structural and electronic properties, graphene-based materials can provide options in several, if not all, of the desired device functionalities for neural implants. | |
| For instance, microtransistors based on CVD graphene can be at the heart of flexible arrays capable of recording over large areas of the brain with very high spatial resolution. To overcome the limitation introduced by the excessive footprint of the large number of connections associated with the high density recording devices, graphene FETs have the additional advantage of enabling facile multiplexing by using flexible technology based on emerging 2D semiconductors, such as MoS2. | |
| With respect to neuro-stimulation, three dimensional or structured graphene films exhibiting high porosity (or surface-to-volume ratio), such as CVD foams or rGO porous films, offer charge injection capacitances similar or superior to those of competing materials. | |
| Furthermore, graphene-related materials (graphene nanoflakes) can be used in hybrid thin films as a doping material to increase the film conductivity and thus to enhance the charge injection of these films. Graphene can also contribute by providing biosensing functionality on neural interfaces based on the reported high sensitivity of detection of electrical and chemical changes. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
