| Nov 04, 2019 | |
3D printing batteries - an overview |
|
| (Nanowerk Spotlight) Energy storage is an integral part of mobile electronics and there is continued demand for ever smaller yet more powerful batteries. Over the years, tremendous efforts have been put into exploring new electrode materials, electrolytes, cell structures, and novel fabrication approaches with the goal of improving the electrochemical performance of batteries, reducing manufacturing cost, and expanding their application. | |
| At the same time, 3D printing is changing our world and the technology is advancing rapidly, quickly becoming the basis of next generation futuristic 3D printed energy architectures where batteries and supercapacitors could be printed in virtually any shape. | |
| Until now, manufacturers have had to design their devices around the size and shape of commercially available batteries, which already take up most of the space in modern electronic devices. Most of them come in cylindrical or rectangular shapes and are optimized for coin cell and/or pouch cells. | |
| Therefore, when a manufacturer is designing a product they must dedicate a certain size and shape to the battery, which could waste space and limit design options. Increasingly this poses a design problem for the coming generations of flexible electronics. | |
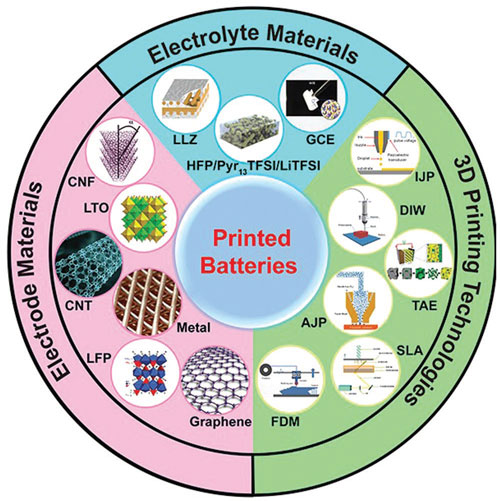
| A review paper in Advanced Functional Materials ("Additive Manufacturing of Batteries") provides a comprehensive overview of 3D-printed batteries via different kinds of printing techniques, including lithography-based 3D printing, template-assisted electrodeposition-based 3D printing, inkjet printing, direct ink writing, fused deposition modeling, and aerosol jet printing. | |
 |
|
| Outline illustration of the review for printed batteries: 3D printing technologies, printed electrode materials, and electrolyte materials. The major 3D printing techniques for manufacturing batteries include lithography-based 3D printing, template-assisted electrodeposition (TAE)-based 3D printing, inkjet printing (IJP), direct ink writing (DIW), fused deposition modeling (FDM), and aerosol jet printing (AJP). Printed electrode materials include graphene, carbon nanotube, carbon nanofiber, and LTO/LFP, and electrolyte materials. (Reprinted with permission by Wiley-VCH Verlag) (click on image to enlarge) | |
| The authors also discuss the working principles, printing process, advantages, and limitations for each 3D printing method and highlight the printing materials for electrodes and electrolytes of the printed batteries. | |
| As an advanced fabrication technique, 3D printing has been increasingly utilized to fabricate complex 3D objects via digitally controlled deposition of phase change and reactive materials and solvent-based inks. | |
| This fabrication method generally begins with the design of a 3D virtual model that is sliced into several 2D horizontal cross sections using special software. By successively printing new 2D layers on top of previous layers, a coherent 3D object can be finally fabricated. | |
 |
|
| This image shows the interlaced stack of electrodes that were 3D-printed layer by layer to create the working anode and cathode of a microbattery that is the size of a grain of sand. For details see: Nanotechnology researchers 3D print microbatteries for implants and tiny robots. | |
| Several types of 3D printing technologies have been developed and are widely utilized today: 1) material extrusion; 2) powder bed fusion; 3) vat photopolymerization; 4) material jetting; 5) binder jetting; 6) sheet lamination; and 7) directed energy deposition. | |
| When it comes to batteries, 3D printing has several significant advantages compared with conventional battery fabrication technologies: 1) enabling the fabrication of desired complex architectures; 2) precise controlling of the shape and thickness of the electrodes; 3) printing solid-state electrolyte with high structure stability and safer operation; 4) potential for low-cost, environmental friendliness, and ease of operation; and 5) possibility of eliminating the steps of device assembly and packaging via direct integration of batteries and other electronics. | |
| Important for future designs, 3D printing is able to fabricate novel 3D-architectured electrodes with larger surface area and higher areal-loading density, providing shorter diffusion pathways and smaller resistance during the ion-transport process so as to improve battery energy density and power density. | |
| With regard to aspects of sustainability and environmental footprint, 3D printing can dramatically reduce material waste and may enable faster production due to the less complex fabrication procedures. | |
| Overall, 3D printing opens new avenues for the rapid fabrication of 3D-structured batteries with complex architectures and high performance. | |
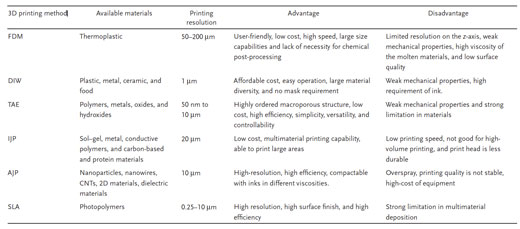
| However, considering the compatibility among the preparation conditions, materials, and processes, not all of the 3D printing technologies and current materials used in conventional batteries are appropriate for manufacturing printed batteries. A qualitative comparison of different 3D printing techniques for printed batteries is shown in the figure below. | |
 |
|
| Comparison of different 3D printing techniques for batteries. (Reprinted with permission by Wiley-VCH Verlag) (click on image to enlarge) | |
| Currently, lithium titanate (LTO) and lithium iron phosphate (LFP) are the most commonly used anode and cathode materials in 3D-printed batteries, exhibiting a minimal volumetric expansion, high rate capability, high stability, and security. | |
| Carbon nanomaterials are another promising material group for the electrode materials used in 3D-printed batteries. Reduced graphene oxide and graphene, for instance, have been used to 3D print ultrafast supercapacitors. | |
| Carbon nanotubes and carbon nanofibers are also very popular materials for printing inks due to their high mechanical strength, high chemical stability, large specific surface area, as well as excellent electrical and thermal properties. | |
| Next to the electrodes, the electrolyte is the most important component for batteries – it serves as catalyst to make a battery conductive by promoting the movement of ions from the cathode to the anode on charge and in reverse on discharge. Electrolyte plays the determining role in electrochemical performance, cycle life, and safety of the battery. | |
| With the continuing advances of 3D printing technologies, the electrolyte of the batteries can also be directly printed so as to reduce the fabrication procedures, fabrication time, and manufacturing cost. | |
| In summary, although much progress has been made in manufacturing 3D-printed batteries, there are still numerous challenges that must be addressed before they can be widely used commercially. | |
| These challenges include the fact that currently there are only a few printable materials, especially active materials, that can be used as inks for 3D printing batteries. Also, most of the current research regarding 3D-printed batteries focuses on the materials of the electrodes and electrolytes. However, despite being important components in batteries, current collectors have rarely been investigated for 3D printing fabrication. | |
| Additionally, for fully 3D-printed batteries, the compatibility of the inks for each component to be printed sequentially and successfully remains a big challenge. | |
| "We expect that with the continuous development of printing techniques and materials, 3D-printed batteries with long-term durability, favorable safety, as well as high energy and power densities will eventually be widely used in many fields," the authors conclude their review. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
