| May 31, 2022 | |
High-performance graphene nanomesh filtration membranes inspired by cell walls |
|
| (Nanowerk Spotlight) Graphene-based membranes have attracted considerable attention as promising candidates for new filtration technologies for filtering out nanoparticles, organic molecules, and even small inorganic salt ions (see our previous Nanowerk Spotlight: "Novel desalination method with nearly perfect salt rejection and high water flux"). | |
| Putting a nature-inspired spin on the fabrication of high-performance graphene membranes for tricky oil/water separations – even in stable emulsions – researchers have demonstrated a graphene nanomesh membrane that possessed high hydrophilicity, super-oleophobicity and low oil adhesion underwater. | |
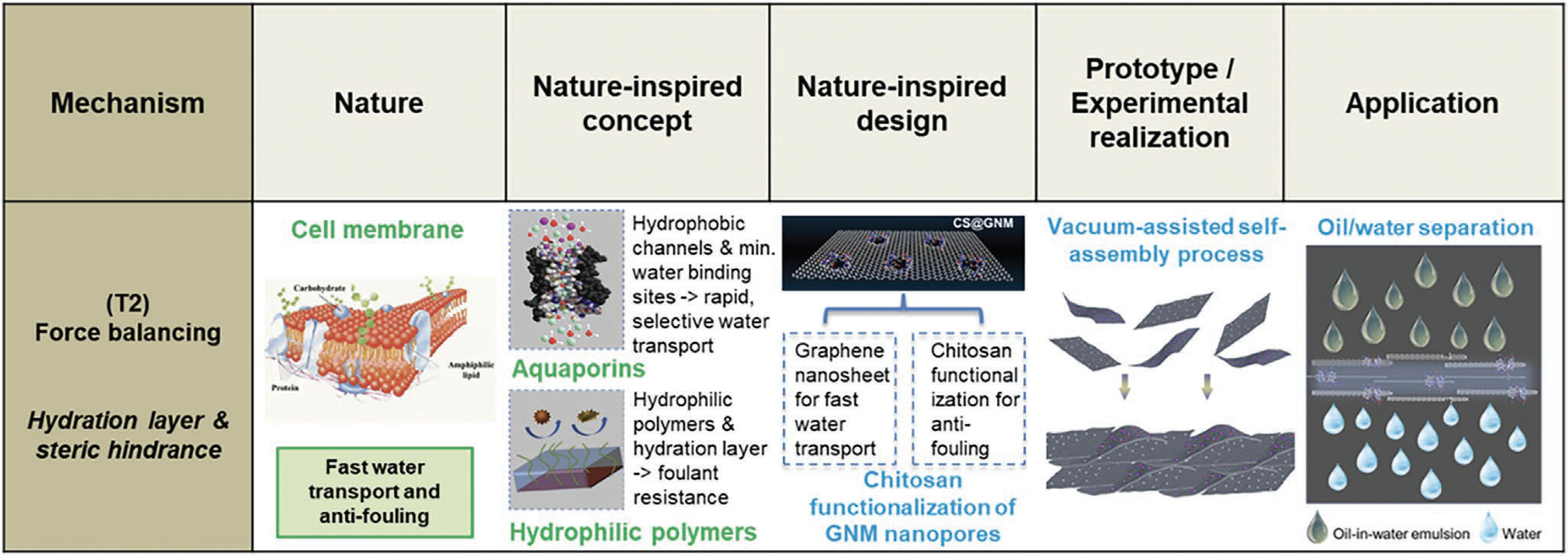
| Reporting their findings in Advanced Functional Materials ("Cell Membrane-Inspired Graphene Nanomesh Membrane for Fast Separation of Oil-in-Water Emulsions"), Marc-Olivier Coppens, Ramsay Memorial Professor in Chemical Engineering at University College London, and Director of UCL's Centre for Nature Inspired Engineering (CNIE), together with first author Yanan Liu, demonstrate graphene nanomesh membranes within a wide pH range at spectacular water permeance (close to 4000 L m–2 h–1 bar–1) under a very low trans-membrane pressure difference. | |
 |
|
| Nature-inspired force balancing mechanisms are used to realize selective high-flux, anti-fouling membranes, as in cell membranes. (Reprinted from doi:10.1002/adfm.202200199 with permission from Wiley-VCH Verlag) (click on image to enlarge) | |
| "We used chitosan-functionalized graphene nanomesh membranes to achieve this superior water flow rate with very high selectivity, resulting in a water recovery ratio of 98.7%," Coppens tells Nanowerk. "The chitosan repels pollutants at the membrane surface and the nanomesh reduces the path length for water molecules, which rapidly slip along and through the graphene layers covered with nano-holes." | |
| Rather than imitating nature out of context, research at CNIE is taking a scientific approach to uncover fundamental mechanisms underlying desirable traits in the natural world. Case in point is this novel membrane technology that leverages the fundamental mechanisms underpinning selective, high water permeation and anti-fouling of biological cell membranes. | |
| "Our nature-inspired chemical engineering (NICE) approach, with its systematic nature-inspired solution methodology, allows us to leverage fundamental mechanisms underpinning desired properties in natural systems – like scalability, efficiency, and resilience – for use within the context of an engineering application," Coppens explains. "We already have demonstrated the success of this approach in projects in fuel cells, sustainable manufacturing, healthcare engineering applications, and more." | |
| The researchers' inspiration for this work comes from the structure of cell membranes, specifically aquaporins. Aquaporins are proteins embedded in cell walls that act as biological channels. They keep cells alive by selectively regulating flow of water, gasses, ions, and other solutes into and out of cells in a manner unmatched by anything humans have made. The reason that aquaporins are so highly effective is that their channel walls repel water (i.e., they are hydrophobic), and that they are quite narrow, having a sub-nanometer diameter. This narrowness forces water to move through the channel in a single-file line at an amazing speed of 3 billion water molecules per second. | |
| Inspired by nature's elegant and highly effective design, the team introduced 'nano-holes' through graphene oxide sheets to create a nanomesh. These nanopores reduce the distance that water has to travel through the membrane and also benefit from the slip along the graphene nanosheets. Combined with the low friction between graphene nanosheets and water molecules, this results in high permeance of almost 4000 L m–2 h–1 bar–1, which is about 260 times the permeance of a graphene oxide membrane. | |
| Fouling is an inevitable problem in membrane separation, where blockages occur in the pores of a membrane, stopping the flow and preventing the membrane from functioning normally. Fouling is an especially severe issue for oil separation technology due to how easily the oil droplets stick onto the membrane surface. | |
| Here, again, nature provided the inspiration. Cell membranes have a natural anti-fouling mechanism thanks to hydrophilic and charged groups that create a hydration layer on the membrane. Having similar functional hydroxyl and amino groups, chitosan has been proposed to functionalize surfaces to be anti-fouling. | |
| Combining these ideas synergistically, the researchers used chitosan with hydrophilic hydroxyl groups and amino groups to modify their graphene nanomesh to increase its hydrophilicity and induce the formation of an anti-fouling hydration layer on the membrane surface. | |
| "Our membrane shows a high permeance and excellent anti-fouling property during separation of surfactant-stabilized oil-in-water emulsions including sunflower oil-in-water emulsion, pump oil-in-water emulsion, octane-in-water emulsion and silicone oil-in-water emulsion," Coppens points out. "It is worth noting that the separation of surfactant-stabilized oil-in water emulsion is much harder than separation of oil-in-water emulsion without the stabilization of surfactant. This is because the size of oil droplets in surfactant-stabilized oil-in-water emulsion (about 200 nm) is smaller than that in surfactant-free oil-in-water emulsion (more than 10 µm), and the oil droplet in the former is much more stable than that in the later." | |
| "Accordingly" he continues, "our cell membrane-inspired graphene nanomesh membrane offers the potential for various applications including petroleum industry and food industry. Meanwhile, our membrane is super-hydrophilic and super-oleophobic underwater, hence, our membrane can let water go through and reject oil droplets in emulsions to produce clean water." | |
| The next stage in this research effort is to scale it up to larger-scale membrane separation modules and to test the long-term stability of the membranes under various practical situations. The researchers also plan to investigate other methods to achieve robust, broad anti-fouling properties of their membranes. | |
| "There is much progress being made in scaling up and reducing the cost of graphene oxide manufacturing, which will help to make this promising methodology cheaper at scale," Coppens concludes. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
