| Aug 02, 2022 | |
A nanofluidic osmotic energy generator |
|
| (Nanowerk Spotlight) In estuaries, where rivers flow into the sea, enormous free-energy dissipation takes place when fresh river water mixes with salty sea water. The release of Gibbs free energy from mixing seawater and fresh water is estimated to be up to 0.8 kW per cubic meter. | |
| This so-called osmotic energy, which normally goes to waste, can be harvested with nanostructured membranes that are put between the two types of water. The salt water pulls fresh water through the membrane and the pressure increases on the salt water side; this pressure increase can be used to produce electrical power with the use of a normal hydroelectric generator. | |
| In 2009, the hydropower company Statcraft built a prototype osmotic power plant in Tufte, Norway. Although a larger commercial plant was supposed to be built, Statcraft closed the plant down four years later due to the negative market outlook at that time. | |
| Nevertheless, researchers estimate that the total amount of energy generated by river water flowing into the oceans each year could theoretically amount to 1.4 to 2.6 TW, which is equivalent to 9-17% of today's global electricity consumption. | |
| Reverse electrodialysis is currently the most promising technology for harvesting osmotic energy – a technology that depends on permselective membranes (i.e., membranes with high ion selectivity and permeability). However, commercial permselective membranes usually face problems of high membrane impedance and poor ion selectivity, resulting in unsatisfactory osmotic energy conversion efficiency. | |
| Boosting osmotic energy harvesting will require new high-performance permselective membranes with low impedance and high ion selectivity. In this context, emerging nanofluidic technologies provide promising platforms for constructing high-performance permselective membranes with both high ion selectivity and high permeability. | |
| The properties at the interfaces of nanochannels play important roles in ion transport during salinity-gradient power conversion. | |
| In recent years, researchers have developed porous materials with intrinsic nano or sub-nanochannels, such as metal–organic frameworks (MOFs) and covalent organic frameworks (COFs) and applied them in various fields including separation and proton exchange membranes. | |
| Especially, COFs are widely used in the field of ion modulation due to their rich ordered nanochannels, tunable pore size and good film-forming ability and they are considered good candidates for nanofluidic osmotic energy harvesting. | |
| However, the preparation of COFs usually based on the solvent thermal synthesis method, which is not only a harsh synthesis process condition but also difficult to form self-standing membranes. | |
| In order to develop self-supporting COF membranes with rich 1D arranged nanochannels for achieving high-efficient osmotic energy harvesting, researchers at Beijing University of Technology now have developed an alternative preparation method. As they report in Advanced Functional Materials ("Ultrathin Self-Standing Covalent Organic Frameworks toward Highly-Efficient Nanofluidic Osmotic Energy Generator"), their ultrathin, self-standing COF has excellent stability at different salt differential concentrations, pH values, and temperatures. | |
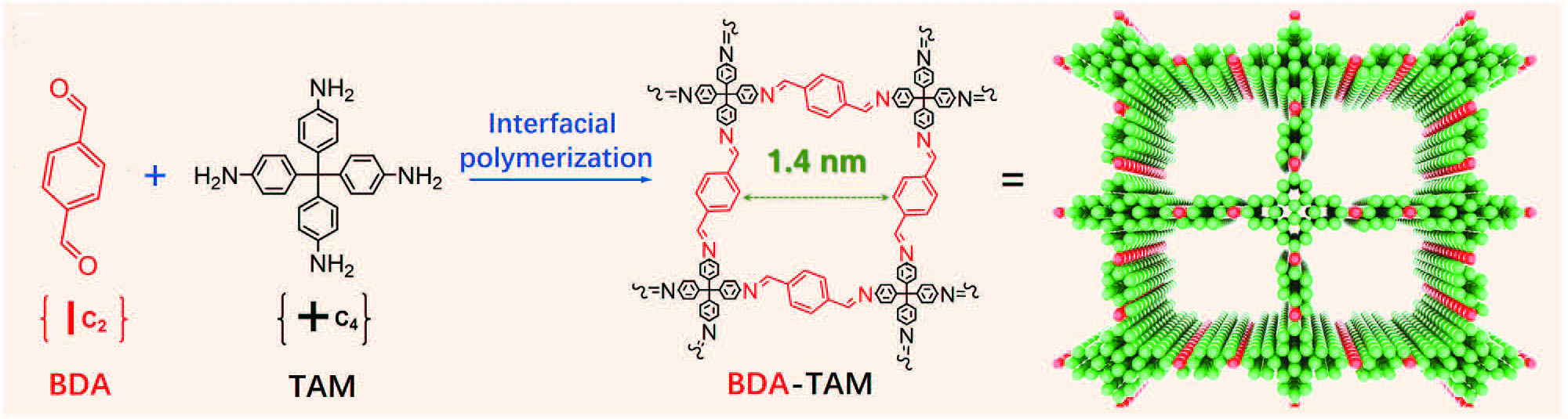
| The team prepared COF membranes via the classical interfacial polymerization based on two monomers of tetrakis(4-aminophenyl)methane (TAM) and terephthalaldehyde (BDA). | |
 |
|
| Schematic illustration of BDA-TAM COFs synthesis process. (Reprinted with permission by Wiley-VCH Verlag) | |
| The resulting ultrathin and large-area membrane features abundant and ordered 1D nanochannels, has a smooth surface and it is self-standing. Its surface area is 166.2 m2 g–1 along with a uniform pore size distribution of ∼1.4 nm and a pore volume of 0.046 cm3 g–1. The thickness of the self-standing COF membrane is as thin as 1.5 µm, which is beneficial for a low membrane resistance and thus promotes the transmembrane cations transport. | |
| When equipped into a nanofluidic osmotic energy generator, this COF nanofluidic membrane can deliver a high output power of 5.31 W m−2 under a 50-fold salinity gradient. Moreover, the COF based osmotic energy generator exhibits excellent stability at different concentrations gradient and pH values and operates well at a wide temperature range. | |
| The authors conclude by pointing out that their work may provide a new insight into the development of high-performance ion perselective membrane for harvesting potential osmotic energy by utilizing COF nanofluidic membranes with rich and rigid 1D nanochannels. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
