| Nov 24, 2023 | |
Unlocking the potential of digital manufacturing for ready-to-use microfluidics |
|
| (Nanowerk Spotlight) For decades, microfluidics has held promise as a revolutionary technology, yet widespread adoption outside research labs has remained elusive. Microfluidic devices enable precise control of tiny volumes of fluid for automated chemical and biological analysis, offering potential breakthroughs in medical testing and diagnostics. However, manufacturing constraints have impeded the field. Clean room facilities and complex peripherals limit access, while static designs reduce versatility. | |
| Now, researchers at McGill University report a breakthrough in overcoming these barriers through digital manufacturing (Advanced Materials, "Digital Manufacturing of Functional Ready-to-Use Microfluidic Systems"). | |
| Microfluidics emerged in the 1990s as microfabrication techniques enabled intricate fluidic circuits to be etched onto chips. By structurally encoding algorithms into interconnected microscopic channels, pumps and valves, complex liquid handling protocols could execute autonomously. This allowed technicians to shrink bulky lab equipment onto disposable credit card-sized devices. | |
| Despite the appeal, microfluidics has faced enduring challenges. Fabrication methods like photolithography demand costly clean room facilities, limiting decentralized production. The need for external pumps and controllers further constrains accessibility and versatility. Once manufactured, devices offer little flexibility or customizability for new applications. Critically, widespread materials like plastics lack required surface chemistry behaviors. These factors have confined microfluidics to academic and corporate labs. | |
| Recent years have seen accelerating interest in applying digital manufacturing, notably additive methods like 3D printing, to microfluidics fabrication. This could enable on-demand, on-site production of bespoke devices. But existing 3D printing approaches still require clean rooms or lack resolution and materials functionality needed for truly integrated microfluidic systems. | |
| The new research demonstrates holistic digital manufacturing of fully functional microfluidic devices in less than 30 minutes on widely accessible 3D printers, sidestepping long-standing barriers. This is achieved through an engineered ink formulation that merges printing compatibility with the capillary effects that enable autonomous microfluidic circuits. Critically, the team shows functioning devices can be printed without any external pumps, controllers or peripheral equipment. | |
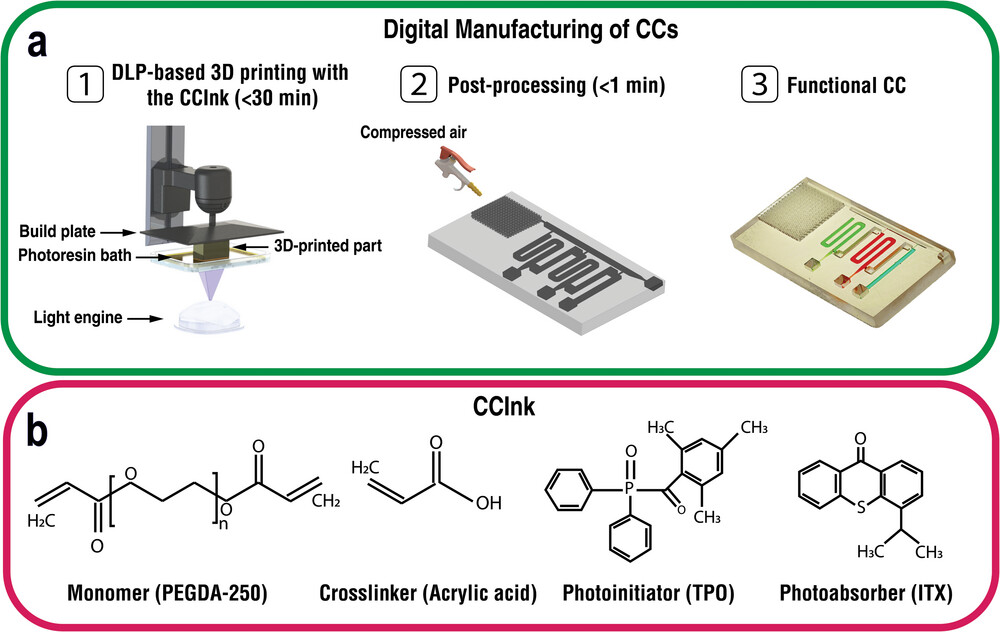 |
|
| Digital manufacturing (DM) of functional capillaric circuits (CCs) using an intrinsically hydrophilic ink (CCInk). a) 1) Additive manufacturing of CCs from a digital design file using a light engine and layer-by-layer vat photopolymerization. 2) Removal of uncured CCInk under a stream of compressed air (<1 min). 3) Functional CC loaded with reagents and primed to execute the structurally encoded (i.e., pre-programmed) capillary flow events following the addition of the triggering solution. b) CCInk composition including polyethyleneglycol diacrylate (PEGDA-250) monomer, acrylic acid (AA) additive to tune hydrophilicity, a photinitiator (TPO), and a photoadsorber (ITX). (Reprinted with permission by Wiley-VCH Verlag) | |
| Building on their pioneering capillaric circuit technology, the researchers incorporated the hydrophilic monomer acrylic acid into a photocurable resin at optimal proportions for balancing print resolution with reliable capillary flow. This permanently enhances surface chemistry behaviors like self-filling channels to sustain liquid movement without external pressure sources. | |
| Several ingenious design innovations overcame further fabrication challenges in translating capillary systems to digital manufacturing. Novel stop valve architectures leverage 3D geometries to halt fluid progression. Circular channel cross-sections prevent problematic bubble trapping. And mathematically generated capillary pumps are structurally integrated to actively pull liquids without paper inserts. | |
| With an engineered material and enhanced component designs in hand, the team demonstrated functioning capillaric circuits embedded inside 3D printed chips. Serpentine channels autonomously wicked solutions while integrated valves sequentially released precise reagent volumes at programmed steps. To validate real-world utility, the researchers ran an immunoassay for COVID antibodies on a printed device just 25 square-centimeters in size. | |
| Notably, even the assay’s nitrocellulose test strip was structurally encoded into the 3D printed main body. By developing the accompanying procedures with no external modules, the total assay time was under 30 minutes. While not yet matching the sensitivity of gold standard testing protocols after sacrificing some wash steps, the fully integrated automation provides a remarkable proof of concept. | |
| This research removes major technology barriers that have long constrained microfluidics and especially autonomous capillary circuits. Compatibility with accessible, mainstream manufacturing methods like widely available light-based 3D printers greatly expands who can produce microfluidic devices. By fabricating functional systems in common plastics without external pumps or controls, overnight printing of application-specific labware becomes feasible. | |
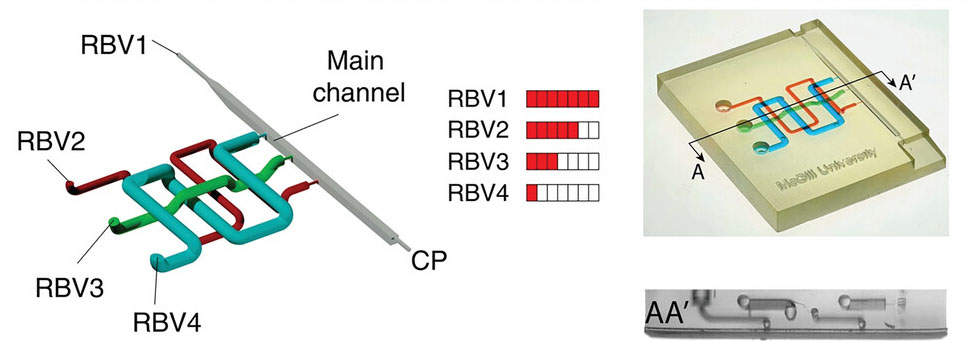 |
|
| 3D-printed capillaric circuits with circular cross-sections and face-centric stop/trigger valves (SVs/TVs) filled with dyed solutions in three reservoirs with 4 retention burst valves (RBVs) with different bursting thresholds for sequential delivery. (Reprinted with permission by Wiley-VCH Verlag) | |
| The minimal skill requirements and modest costs further promote decentralized production and customization. And the engineered hydrophilic resin formulation provides durable surface functionality. Altogether this unlocks microfluidics potential in fields from medical diagnostics to pharmaceutical testing, water quality analysis to agriculture. | |
| Standing on the achievements reported here, researchers next aim to continue enhancing design resolution, reliability and integration. Optimizing geometries and surface treatments may bolster valve performance and pumping pressure limits. Tighter feature size tolerances could increase channel density. And alternative readout methods like embedding reactive particle beds to detect target analytes show promise for reducing or removing external labware like test strips. | |
| Combined with online sharing of printable device templates, this critical advance ushers in an era where multifunctional microfluidic systems can propagate through digital manufacturing as easily as documents flow from computer file to office printer. Just as word processing revolutionized information technology by enabling instant editing and printing of custom text, these printable microfluidic “apps” stand to spark a community-driven revolution in scientific hardware. The future looks fluid. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
