 Nanoparticles are valuable and useful in many products, but according to a new study, they can also damage our cells. Researchers are concerned about the effect of lifelong exposure to the human organism.
Nanoparticles are valuable and useful in many products, but according to a new study, they can also damage our cells. Researchers are concerned about the effect of lifelong exposure to the human organism.
Oct 1st, 2020
Read more
 Researchers have successfully overcome a limiting problem with stabilizing the best-performing formulation of metal-halide perovskite films, a key player in a range of applications, including solar cells.
Researchers have successfully overcome a limiting problem with stabilizing the best-performing formulation of metal-halide perovskite films, a key player in a range of applications, including solar cells.
Oct 1st, 2020
Read more
 Researchers have developed accurate radiation sources that are expected to lead to breakthroughs in medical imaging and other areas.
Researchers have developed accurate radiation sources that are expected to lead to breakthroughs in medical imaging and other areas.
Oct 1st, 2020
Read more
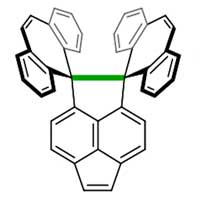 Researchers have successfully demonstrated that carbon-carbon (C-C) covalent bonds expand and contract flexibly in response to light and heat. This unexpected flexibility of C-C bonds could confer new properties to organic compounds.
Researchers have successfully demonstrated that carbon-carbon (C-C) covalent bonds expand and contract flexibly in response to light and heat. This unexpected flexibility of C-C bonds could confer new properties to organic compounds.
Oct 1st, 2020
Read more
 Researchers developed a new class of broadband solid state light emitter in the short-wave infrared that could be miniaturized, integrated with CMOS technology and used for many applications including food inspection, health or safety.
Researchers developed a new class of broadband solid state light emitter in the short-wave infrared that could be miniaturized, integrated with CMOS technology and used for many applications including food inspection, health or safety.
Oct 1st, 2020
Read more
 Researchers have developed a graphene-based bolometer that can detect microwave photons at extremely high sensitivities and with fast time responses.
Researchers have developed a graphene-based bolometer that can detect microwave photons at extremely high sensitivities and with fast time responses.
Oct 1st, 2020
Read more
 What happens to the nanoparticles when they are injected into the bloodstream, for example, to destroy solid tumours? Researchers are now ready to tackle such a challenging question using zebrafish embryos as a new study model in nanomedicine and nanotoxicology.
What happens to the nanoparticles when they are injected into the bloodstream, for example, to destroy solid tumours? Researchers are now ready to tackle such a challenging question using zebrafish embryos as a new study model in nanomedicine and nanotoxicology.
Sep 30th, 2020
Read more
 New research findings show potential for graphene bolometers to become a game-changer for quantum technology.
New research findings show potential for graphene bolometers to become a game-changer for quantum technology.
Sep 30th, 2020
Read more
 Scientists developed a new method and discover new features of primary cilia - little understood antenna-like structures protruding from cells.
Scientists developed a new method and discover new features of primary cilia - little understood antenna-like structures protruding from cells.
Sep 30th, 2020
Read more
 Researchers have synthesized a new porous material that enables and guides the degradation of compounds analogous to nerve agents used in chemical warfare. This material will make it possible to capture and degrade this type of compounds that until now could not be eliminated.
Researchers have synthesized a new porous material that enables and guides the degradation of compounds analogous to nerve agents used in chemical warfare. This material will make it possible to capture and degrade this type of compounds that until now could not be eliminated.
Sep 30th, 2020
Read more
 A self-erasing chip for security and anti-counterfeit technologies.
A self-erasing chip for security and anti-counterfeit technologies.
Sep 30th, 2020
Read more
 Dual-mode thermal rectification could be a game changer for a range of industrial and medical applications.
Dual-mode thermal rectification could be a game changer for a range of industrial and medical applications.
Sep 30th, 2020
Read more
 Researchers devlop innovative coating for bipolar plates in fuel cells.
Researchers devlop innovative coating for bipolar plates in fuel cells.
Sep 30th, 2020
Read more
 For the first time, researchers have shown that highly ordered copper thin films can be crystallized directly on a one-molecule-thick layer of organic material rather than on the inorganic substrates that have been used for years.
For the first time, researchers have shown that highly ordered copper thin films can be crystallized directly on a one-molecule-thick layer of organic material rather than on the inorganic substrates that have been used for years.
Sep 30th, 2020
Read more
 A nanocoating designed to allow air filters to capture airborne or aerosolized droplets of the virus that causes COVID-19.
A nanocoating designed to allow air filters to capture airborne or aerosolized droplets of the virus that causes COVID-19.
Sep 29th, 2020
Read more
 Scientists typically prefer to work with ordered systems. However, a diverse team of physicists and biophysicists found that individual light-harvesting nanotubes with disordered molecular structures still transport light energy in the same way.
Scientists typically prefer to work with ordered systems. However, a diverse team of physicists and biophysicists found that individual light-harvesting nanotubes with disordered molecular structures still transport light energy in the same way.
Sep 29th, 2020
Read more
 Nanoparticles are valuable and useful in many products, but according to a new study, they can also damage our cells. Researchers are concerned about the effect of lifelong exposure to the human organism.
Nanoparticles are valuable and useful in many products, but according to a new study, they can also damage our cells. Researchers are concerned about the effect of lifelong exposure to the human organism.















 Subscribe to our Nanotechnology News feed
Subscribe to our Nanotechnology News feed