Cosmic rays may soon stymie quantum computing
 Building quantum computers underground or designing radiation-proof qubits may be needed, researchers find.
Building quantum computers underground or designing radiation-proof qubits may be needed, researchers find.
Aug 28th, 2020
Read more
 Building quantum computers underground or designing radiation-proof qubits may be needed, researchers find.
Building quantum computers underground or designing radiation-proof qubits may be needed, researchers find.
Aug 28th, 2020
Read more Researchers have developed a stretchable, wearable gas sensor for environmental sensing.
Researchers have developed a stretchable, wearable gas sensor for environmental sensing.
Aug 28th, 2020
Read more Extremely energy-efficient artificial intelligence is now closer to reality after researchers found a way to improve the accuracy of a brain-inspired computing system.
Extremely energy-efficient artificial intelligence is now closer to reality after researchers found a way to improve the accuracy of a brain-inspired computing system.
Aug 27th, 2020
Read more Using a material widely adopted by photonics researchers, researchers have created the smallest electro-optical modulator yet. The modulator is a key component of a photonics-based chip, controlling how light moves through its circuits.
Using a material widely adopted by photonics researchers, researchers have created the smallest electro-optical modulator yet. The modulator is a key component of a photonics-based chip, controlling how light moves through its circuits.
Aug 27th, 2020
Read more Chemists have successfully tied a series of microscopic knots using individual molecules for the first time, ushering in the advent of a form of nano-scale weaving which could create a new generation of advanced materials.
Chemists have successfully tied a series of microscopic knots using individual molecules for the first time, ushering in the advent of a form of nano-scale weaving which could create a new generation of advanced materials.
Aug 27th, 2020
Read more By mixing electronic resonances in semiconductor nanostructures with the photon field of microresonators, researchers designed a stained mirror that bleaches more easily than ever and could make terahertz lasers ultrafast.
By mixing electronic resonances in semiconductor nanostructures with the photon field of microresonators, researchers designed a stained mirror that bleaches more easily than ever and could make terahertz lasers ultrafast.
Aug 27th, 2020
Read more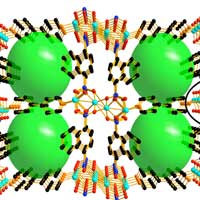 The materials belong to the class of metal-organic frameworks, or MOFs for short, and could open up new opportunities for energy storage.
The materials belong to the class of metal-organic frameworks, or MOFs for short, and could open up new opportunities for energy storage.
Aug 27th, 2020
Read more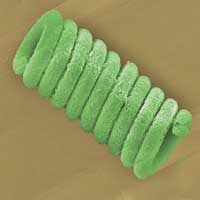 Scientists have equipped these humble unicellular organisms with semiconductor coatings to create mini power plants, which supply biofuels and are photocatalytically active in sunlight.
Scientists have equipped these humble unicellular organisms with semiconductor coatings to create mini power plants, which supply biofuels and are photocatalytically active in sunlight.
Aug 27th, 2020
Read more The BIOMAP process uses microparticles of varying sizes. The particles are applied to a surface, covering it and form-fitting all the crevices, gaps and patterns through the autonomous processes of self-filtration, capillary pressure and evaporation.
The BIOMAP process uses microparticles of varying sizes. The particles are applied to a surface, covering it and form-fitting all the crevices, gaps and patterns through the autonomous processes of self-filtration, capillary pressure and evaporation.
Aug 27th, 2020
Read more Technique may promote practical use of high-performance magnetoresistive devices comprising half-metallic heusler alloys.
Technique may promote practical use of high-performance magnetoresistive devices comprising half-metallic heusler alloys.
Aug 27th, 2020
Read more New spectroscopic technique for studying nanostructures demonstrates that stibnite nanodots can act as high-optical-quality waveguides and are promising candidates as photoswitchable materials for future applications.
New spectroscopic technique for studying nanostructures demonstrates that stibnite nanodots can act as high-optical-quality waveguides and are promising candidates as photoswitchable materials for future applications.
Aug 26th, 2020
Read more The study found that flexibility, as well as density, in the bone nanostructure is an important factor in assessing how likely someone is to suffer fractures.
The study found that flexibility, as well as density, in the bone nanostructure is an important factor in assessing how likely someone is to suffer fractures.
Aug 26th, 2020
Read more Researchers create portable lab-on-a-chip that could detect many contaminants.
Researchers create portable lab-on-a-chip that could detect many contaminants.
Aug 26th, 2020
Read more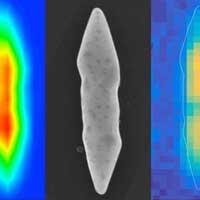 Gold nanoparticles can be better catalysts if their atoms are packed differently from the conventional arrangement. This alternate inner structure leads to different inherent properties.
Gold nanoparticles can be better catalysts if their atoms are packed differently from the conventional arrangement. This alternate inner structure leads to different inherent properties.
Aug 26th, 2020
Read more Researchers have developed the world's first afterglow-magnetic nanoparticles. The patented invention is designed for various applications including cancer detection in medicine and the detection of fine particulates in living organisms.
Researchers have developed the world's first afterglow-magnetic nanoparticles. The patented invention is designed for various applications including cancer detection in medicine and the detection of fine particulates in living organisms.
Aug 26th, 2020
Read more Researchers have created a machine-learning algorithm that can characterise graphene properties and quality within 14 minutes.
Researchers have created a machine-learning algorithm that can characterise graphene properties and quality within 14 minutes.
Aug 26th, 2020
Read more