At the edge of chaos, powerful new electronics could be created
 Study shows how ferroic materials could be used to create adaptable neuromorphic electronics.
Study shows how ferroic materials could be used to create adaptable neuromorphic electronics.
Sep 3rd, 2019
Read more
 Study shows how ferroic materials could be used to create adaptable neuromorphic electronics.
Study shows how ferroic materials could be used to create adaptable neuromorphic electronics.
Sep 3rd, 2019
Read more In what could be a breakthrough for body sensor and navigation technologies, researchers have developed the smallest accelerometer yet reported, using the highly conductive nanomaterial, graphene.
In what could be a breakthrough for body sensor and navigation technologies, researchers have developed the smallest accelerometer yet reported, using the highly conductive nanomaterial, graphene.
Sep 3rd, 2019
Read more An electrocatalysis reactor recycles carbon dioxide to produce pure liquid fuel solutions using electricity. The scientists behind the invention hope it will become an efficient and profitable way to reuse the greenhouse gas and keep it out of the atmosphere.
An electrocatalysis reactor recycles carbon dioxide to produce pure liquid fuel solutions using electricity. The scientists behind the invention hope it will become an efficient and profitable way to reuse the greenhouse gas and keep it out of the atmosphere.
Sep 3rd, 2019
Read more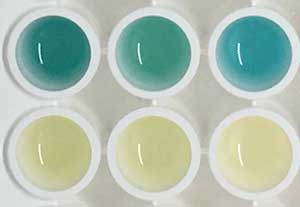 A simple and sensitive urine test has produced a colour change in urine to signal growing tumours in mice.
A simple and sensitive urine test has produced a colour change in urine to signal growing tumours in mice.
Sep 2nd, 2019
Read more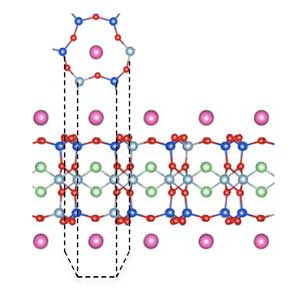 Researchers discovered that atomically thin micas - the name given to a type of common mineral found in soil - are excellent proton conductors. This surprising result is important for the use of 2D materials in applications such as fuel cells and other hydrogen-related technologies.
Researchers discovered that atomically thin micas - the name given to a type of common mineral found in soil - are excellent proton conductors. This surprising result is important for the use of 2D materials in applications such as fuel cells and other hydrogen-related technologies.
Sep 2nd, 2019
Read more Chemists have significantly improved the experimental determination of the chirality or 'handedness' of molecules using vibrational circular dichroism (VCD) spectroscopy.
Chemists have significantly improved the experimental determination of the chirality or 'handedness' of molecules using vibrational circular dichroism (VCD) spectroscopy.
Sep 2nd, 2019
Read more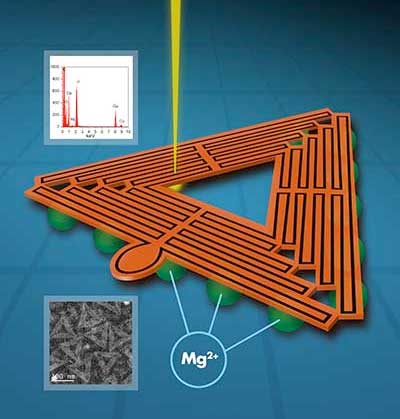 Scientists are now able to see greater details of DNA origami nanostructures, which will lead to a greater understanding and control of their assembly for future applications.
Scientists are now able to see greater details of DNA origami nanostructures, which will lead to a greater understanding and control of their assembly for future applications.
Sep 2nd, 2019
Read more Incorporating individual metal atoms into a surface in the right way allows their chemical behavior to be adapted. This makes new, better catalysts possible.
Incorporating individual metal atoms into a surface in the right way allows their chemical behavior to be adapted. This makes new, better catalysts possible.
Sep 2nd, 2019
Read more Cell particles move more quickly through a crowded cellular environment when the crowding molecules are non-uniformly distributed.
Cell particles move more quickly through a crowded cellular environment when the crowding molecules are non-uniformly distributed.
Aug 31st, 2019
Read more Low-cost piezoelectric films produce voltage when under strain, could be used for flexible electronic components and more.
Low-cost piezoelectric films produce voltage when under strain, could be used for flexible electronic components and more.
Aug 30th, 2019
Read more Scientists suggested a model describe the distribution of heat in ultrapure crystals at the atomic level.
Scientists suggested a model describe the distribution of heat in ultrapure crystals at the atomic level.
Aug 30th, 2019
Read more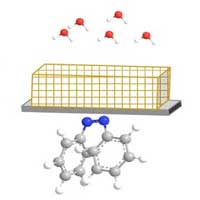 Researchers have developed new materials that can absorb and release small molecules, and it can be controlled by shining light on it.
Researchers have developed new materials that can absorb and release small molecules, and it can be controlled by shining light on it.
Aug 30th, 2019
Read more Scientists have investigated the suitability of a system based on nanodiamonds as diagnostic and therapeutic method for brain diseases.
Scientists have investigated the suitability of a system based on nanodiamonds as diagnostic and therapeutic method for brain diseases.
Aug 30th, 2019
Read more Researchers have developed a method for assembling these complex molecules from a small number of segments.
Researchers have developed a method for assembling these complex molecules from a small number of segments.
Aug 30th, 2019
Read more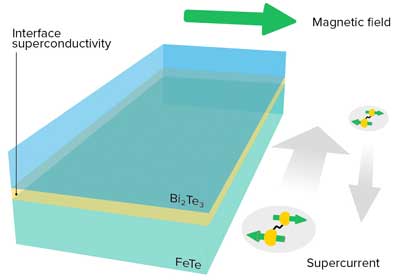 A composite material reveals the relationship between superconductivity and exotic electronic states that could be useful in quantum computers.
A composite material reveals the relationship between superconductivity and exotic electronic states that could be useful in quantum computers.
Aug 30th, 2019
Read more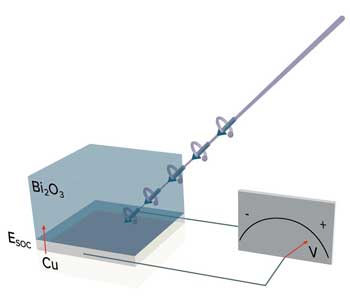 The interface between a metal and an oxide creates the right conditions for optically controlling electrons.
The interface between a metal and an oxide creates the right conditions for optically controlling electrons.
Aug 30th, 2019
Read more