New spin state discovered near absolute zero
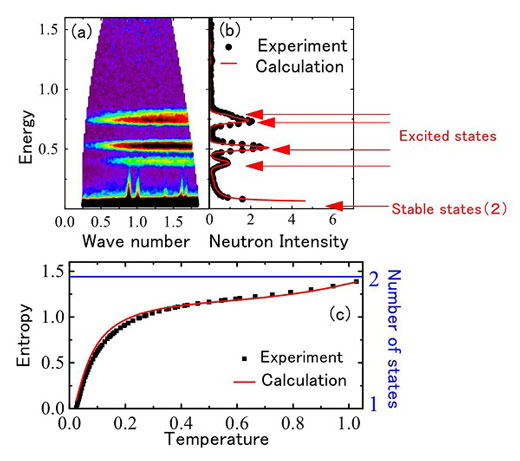 Tetrahedral spins challenge third law of thermodynamics.
Tetrahedral spins challenge third law of thermodynamics.
Jun 21st, 2016
Read more
 Tetrahedral spins challenge third law of thermodynamics.
Tetrahedral spins challenge third law of thermodynamics.
Jun 21st, 2016
Read more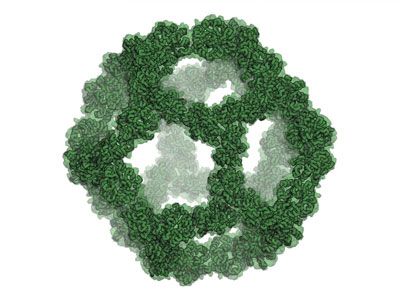 Made-to-order nanocages open possibilities of shipping cargo into living cells, building small chemical reactors.
Made-to-order nanocages open possibilities of shipping cargo into living cells, building small chemical reactors.
Jun 21st, 2016
Read more Researchers took a counterintuitive approach by taking advantage of optical distortions that they typically try to eliminate.
Researchers took a counterintuitive approach by taking advantage of optical distortions that they typically try to eliminate.
Jun 20th, 2016
Read more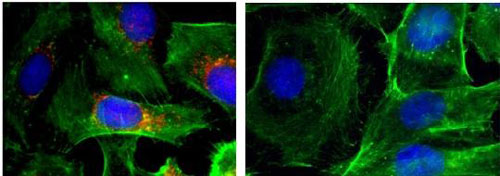 Platform designed to deliver nanotherapy effective in preclinical models of metastatic anaplastic thyroid cancer.
Platform designed to deliver nanotherapy effective in preclinical models of metastatic anaplastic thyroid cancer.
Jun 20th, 2016
Read more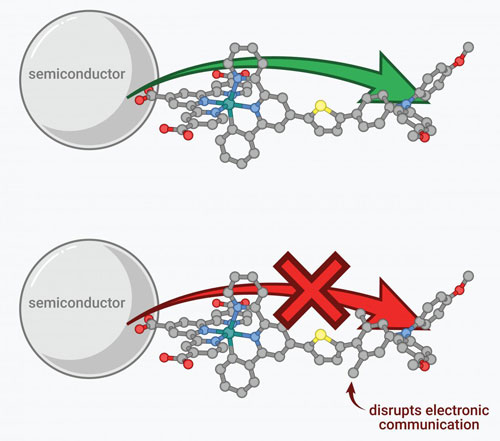 Researchers have discovered a new way to optimize electron transfer in semi-conductors used in solar fuel solutions.
Researchers have discovered a new way to optimize electron transfer in semi-conductors used in solar fuel solutions.
Jun 20th, 2016
Read more DNA nanowire improved by altering sequences.
DNA nanowire improved by altering sequences.
Jun 20th, 2016
Read more A Franco-Japanese research group has developed a new 'brute force' technique to test thousands of biochemical reactions at once and quickly home in on the range of conditions where they work best.
A Franco-Japanese research group has developed a new 'brute force' technique to test thousands of biochemical reactions at once and quickly home in on the range of conditions where they work best.
Jun 20th, 2016
Read more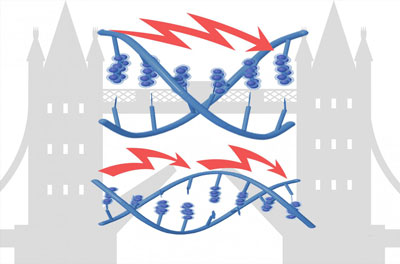 Scientists have been able to understand and manipulate DNA to more finely tune the flow of electricity through it.
Scientists have been able to understand and manipulate DNA to more finely tune the flow of electricity through it.
Jun 20th, 2016
Read more Researchers have fabricated a solution-processed, semi-transparent solar cell based on AgBiS2 nanocrystals, a material that consists of non-toxic, earth-abundant elements, produced in ambient conditions at low temperatures.
Researchers have fabricated a solution-processed, semi-transparent solar cell based on AgBiS2 nanocrystals, a material that consists of non-toxic, earth-abundant elements, produced in ambient conditions at low temperatures.
Jun 20th, 2016
Read more The flexible photovoltaics could power wearable electronics.
The flexible photovoltaics could power wearable electronics.
Jun 20th, 2016
Read more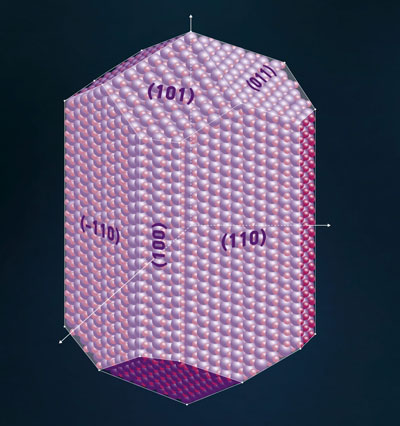 Using numerical modelling, researchers from Russia, the US, and China have discovered previously unknown features of rutile TiO2, which is a promising photocatalyst.
Using numerical modelling, researchers from Russia, the US, and China have discovered previously unknown features of rutile TiO2, which is a promising photocatalyst.
Jun 20th, 2016
Read more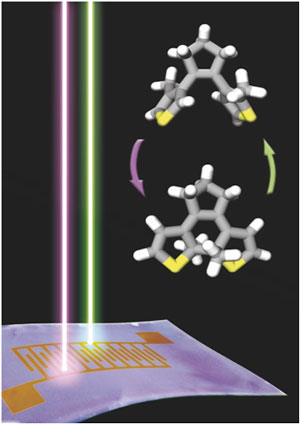 Researchers have shown that a carefully chosen blend of a small photoswitchable molecule and a semiconducting polymer can be used to fabricate high-performance memory devices that can be written and erased by light.
Researchers have shown that a carefully chosen blend of a small photoswitchable molecule and a semiconducting polymer can be used to fabricate high-performance memory devices that can be written and erased by light.
Jun 20th, 2016
Read moreResearchers develop an organic nanofiber based artificial synapse that emulates both important functions and energy consumption of biological ones: Implication of potential use in artificial intelligence computing.
Jun 20th, 2016
Read more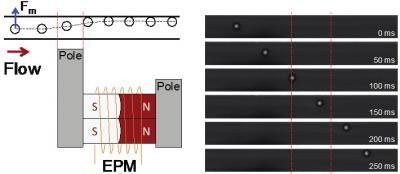 Researchers have demonstrated magnetic actuation of microfluidic water droplets using miniature EPMs and oil-based ferrofluids.
Researchers have demonstrated magnetic actuation of microfluidic water droplets using miniature EPMs and oil-based ferrofluids.
Jun 20th, 2016
Read more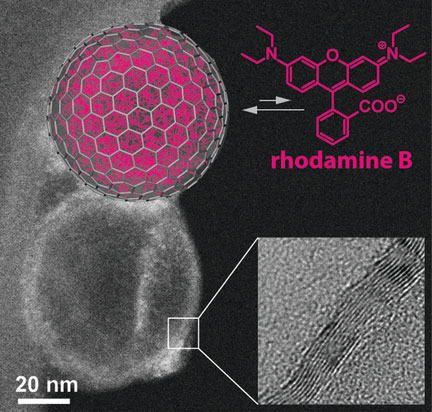 Scientists have introduced a new variety of nanocapsule: hollow carbon bubbles made from three to four graphitic layers of carbon atoms.
Scientists have introduced a new variety of nanocapsule: hollow carbon bubbles made from three to four graphitic layers of carbon atoms.
Jun 20th, 2016
Read more To accurately determine relative energies of the electrons and signatures of the spin-orbit interaction in an oxide material, researchers performed resonant inelastic x-ray scattering (RIXS). The results reveal divergent behavior than found from RIXS measurements in related iridates that are candidates for developing high-temperature superconductors.
To accurately determine relative energies of the electrons and signatures of the spin-orbit interaction in an oxide material, researchers performed resonant inelastic x-ray scattering (RIXS). The results reveal divergent behavior than found from RIXS measurements in related iridates that are candidates for developing high-temperature superconductors.
Jun 19th, 2016
Read more