| Posted: Jun 19, 2018 |
Nanocrystals impact stem cell fate during bone formation
(Nanowerk News) Scientists from the University of Freiburg and the University of Basel identified a master regulator for bone regeneration. Prasad Shastri, Professor of Biofunctional Macromolecular Chemistry at the Institute for Macromolecular Chemistry and Professor of Cell Signalling Environments in the Cluster of Excellence BIOSS Centre for Biological Signalling Studies at the University of Freiburg led the study. The team published the results in the journal Proceedings of the National Academy of Sciences ("Hyperstimulation of CaSR in human MSCs by biomimetic apatite inhibits endochondral ossification via temporal down-regulation of PTH1R").
|
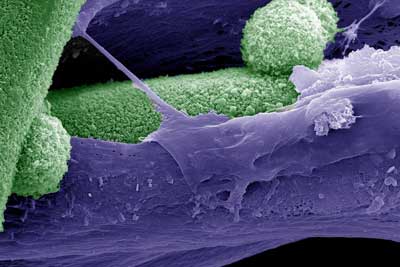 |
| The image is false colored scanning electron micrograph, where mineral phase (biomimetic bone like hydroxyapatite) is denoted by green and the matrix deposited by mesenchymal stem cells is shown in purple. (Image: Melika Sarem, Vincent Ahmadi and V. Prasad Shastri)
|
|
Bone tissue is a nano-composite: The inorganic mineral phase so called hydroxyapatite confers the bone stability, and these calcium-containing nano crystals are dispersed in an organic framework consisting of the protein collagen.
|
|
Although bone tissue rejuvenates itself throughout one’s life, inducing damaged bone to repair has proved challenging. The destruction of unhealthy bone is a necessary step in the saga of new bone formation. During this destruction of bone, many biomolecules that are stored in the organic matrix are released and they play a role in the bone restoration process by coaxing mesenchymal stem cells (MSCs) that reside in the bone marrow – to actively form bone.
|
|
This step presents a crossroad of sorts in the process of bone regeneration in that, MSCs can become bone cells and deposit bone or become cartilage cells and deposit a cartilage matrix (the callus) which then is transformed into bone. How the accompanying degradation of the hydroxyapatite matrix affects this crossroad has remained a mystery of sorts.
|
|
Using a biomimetic bone-like mineral phase developed in the laboratory of Shastri (Advanced Materials, "Disordered Conformation with Low Pii Helix in Phosphoproteins Orchestrates Biomimetic Apatite Formation"), the team of scientists have unraveled that the bone mineral phase is a key “decision maker” in bone formation.
|
|
In the study post-doctoral associate Dr. Melika Sarem from the group of Shastri, in collaboration with the research group of Prof. Ivan Martin at the Department of Biomedicine (University Hospital Basel, University of Basel), have discovered that the mineral phase of bone can stimulate a receptor called extracellular calcium sensing receptor (CaSR), a protein that senses calcium outside the cells and is expressed by MSCs. Over stimulation of CaSR forces MSCs to directly form bone as opposed to via a cartilage step.
|
|
They further report that interfering with signaling via CaSR can completely shut down the formation of bone in vivo. However, in a twist to this molecular dance, they have found that stimulating parathyroid hormone-1 receptor (PTH1R), the key regulator of calcium ion homeostasis, can rescue MSCs from the clutches of CaSR and promote the formation of bone via a cartilage intermediate.
|
|
“Our discovery offers new insights into how bone mineral phase can dictate new bone formation” says Prof. Shastri. The findings of our study have huge implication for designing novel implant surface for bone regeneration” adds Dr. Sarem.
|
|
In diseases such as osteoporosis, bone is degraded with very little renewal of the lost bone. “Our study places CaSR squarely at the middle of the bone regeneration paradigm and we can now say that it is a master regulator of bone formation and this might explain why osteoporotic patients have a hard time healing their fractures” says Shastri.
|

