| Jun 11, 2021 |
New family of atomic-thin electride materials discovered
(Nanowerk News) An exploratory investigation into the behavior of materials with desirable electric properties resulted in the discovery of a structural phase of two-dimensional (2D) materials. The new family of materials are electrides, wherein electrons occupy a space usually reserved for atoms or ions instead of orbiting the nucleus of an atom or ion. The stable, low-energy, tunable materials could have potential applications in nanotechnologies.
|
|
The international research team, led by Hannes Raebiger, associate professor in the Department of Physics at Yokohama National University in Japan, published their results as frontispiece in Advanced Functional Materials ("MXene Phase with C3 Structure Unit: A Family of 2D Electrides").
|
|
Initially, the team set out to better understand the fundamental properties of a 2D system known as Sc2CO2. Containing two atoms of metallic scandium, one atom of carbon and two atoms of oxygens, the system belongs to a family of chemical compounds collectively referred to as MXenes. They are typically composed of a carbon or nitrogen layer one atom thick sandwiched between metal layers, dotted with oxygen or fluorine atoms.
|
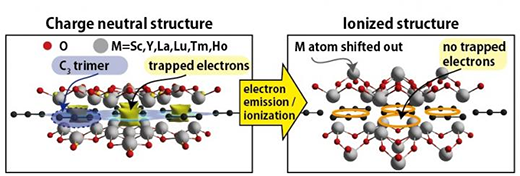 |
| Yellow isosurfaces on left panel indicate electrons localized in-between the C3 trimers. Ionized structure on the right has no trapped electrons, and some of the M atoms have been largely displaced. This displacement of the M atoms again significantly stabilizes the ionized structure. (IUmage: Soungmin Bae and Hannes Raebiger)
(click on image to enlarge)
|
|
The researchers were particularly interested in MXene Sc2CO2 due to the predictions that, when structured into a hexagonal phase, the system would have desired electrical properties.
|
|
"Despite these fascinating predictions of hexagonal phases of Sc2CO2, we are not aware of its successful fabrication as of yet," said Soungmin Bae, first author and researcher in the Department of Physics at Yokohama National University. "Analyzing its fundamental properties, we discovered a completely new structural phase."
|
|
The new structural phase results in new electride materials. The atomic-thin 2D structural phase is described as tiled shapes forming the central carbon plane. The previously predicted shape was a hexagon, with a carbon atom at every vertex and one in the middle. The new materials have a rhombus-like shape, with electrons at the vertices and a carbon trimer -- three carbon atoms in a row -- in the middle.
|
|
"Carbon is one of the most common materials on our planet, and quite important for living beings, but it is hardly ever found as trimers," Raebiger said. "The closest place where carbon trimers are typically found is interstellar space."
|
|
The overall shape is less symmetric than the previously described hexagonal structure, but it is more symmetric with regard to the central plane. This structure offers unique characteristics due to the appearance of the new family of electrides, according to Raebiger.
|
|
"Electrides contain electrons as a structural unit and often are extremely good electrical conductors," Raebiger said. "The present family of electrides are insulators, and while most insulators can be made conductive by adding or removing electrons, these materials simply become more insulating."
|
|
MXenes are particularly attractive as a material, because they can be reconfigured with other metallic elements to offer a cornucopia of properties, including tunable conductivity, various forms of magnetism, and/or accelerate chemical reactions as catalysts. On top of this, they are ultra-thin sheets only a few atoms thick, that is, 2D materials. The newly discovered electrides have electrons in lattice voids between atoms and ions, which can be readily emitted into surrounding space, such as the electron sources for large particle accelerators, as well as be borrowed to catalyze a specifically desired chemical reaction.
|
|
"We made this discovery because we wanted to understand how these materials work better," Bae said. "If you encounter something you don't understand, dig deeper."
|

