| Dec 09, 2023 |
Empowering optical tweezers with 'biometric eyes'
(Nanowerk News) Researchers have long sought to develop versatile tools that allow precise manipulation and identification of DNA and other biological nanoparticles. Optical tweezers, which use lasers to trap small particles, offer contactless control but have lacked the capability to recognize specific DNA sequences.
|
|
At the same time, CRISPR gene editing has emerged as an ultra-sensitive detection method but requires enhancement and integration with nanotechnology. Now, researchers from Shenzhen University and the Chinese University of Hong Kong have combined CRISPR with optothermal tweezers in an innovation that overcomes prior limitations.
|
|
In a new study published in Light: Science & Applications ("CRISPR-powered optothermal nanotweezers: Diverse bio-nanoparticle manipulation and single nucleotide identification"), the team presents CRISPR-powered optothermal nanotweezers (CRONT) – a platform that offers simultaneous trapping and identification of DNA with high accuracy. This breakthrough paves the way for advances in point-of-care testing, biosensing, and fundamental biophysics research.
|
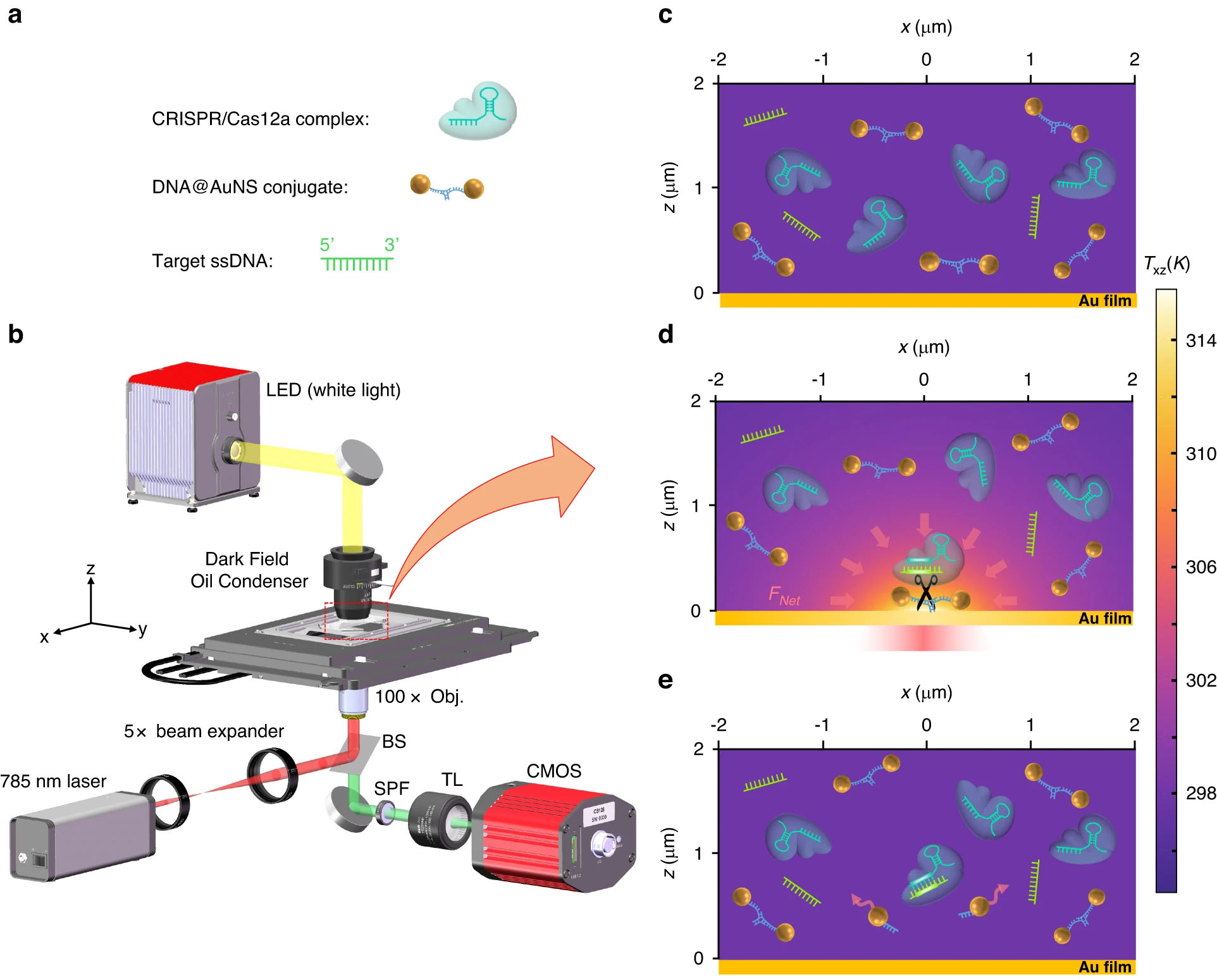 |
| Working principle of CRISPR-powered optothermal nanotweezers. a The diagrammatic sketch of the three components in the solution: DNA@AuNS conjugate, CRISPR/Cas12a complex, and target ssDNA. b Optical setup, the BS, SPF, and TL are beam splitter, short pass filter, and tube lens (f=200 mm), respectively. Additional details of the setup are provided in the Materials and Methods section. c Dispersion of the three components in the solution without optical heating. d Optothermal net force (FNet) induced migration and DNA@AuNS conjugate cleavage upon optical heating, the heating laser power is 0.5 mW. e Observation of the cleavage after the optical heating is switched off. (© Light: Science & Applications)
|
|
Optical tweezers have become an invaluable technique since their invention in 1986, providing unparalleled spatiotemporal control over microscopic particles. But they rely on momentum transfer from a tightly focused, high-powered laser, limiting the materials that can be trapped and introducing photodamage risks from local heating.
|
|
Optothermal tweezers were introduced 15 years ago as a promising alternative with much lower intensities. They harness microscale temperature gradients and fluid flow near an illuminated light-absorbing surface to manipulate particles through thermodiffusion and convective forces. But until now, both traditional optical tweezers and optothermal techniques were unable to directly identify biological particles using optical signals. That capability has only been achieved through cumbersome fluorescence tagging methods.
|
|
In parallel, CRISPR diagnostics have become a groundbreaking approach to detect nucleic acid sequences with extreme specificity down to single base pairs. However, translating benchtop assays into point-of-care devices remains challenging due to the extensive sample preparation and target amplification needed. The Shenzhen-CUHK team realized the enormous potential of fusing CRISPR sensing with the precise manipulation of optothermal tweezers in one platform.
|
|
The CRONT system uses a thin gold film to generate heated spots just above room temperature upon illumination with a standard 785 nm laser pointer. This creates localized convection currents and thermodiffusion that can selectively trap over 50 distinct DNA sequences and nanoparticles functionalized with DNA. Once trapped, sequences are identified in-situ by introducing CRISPR complexes that cleave target DNA strands, observed in real-time as the diffraction pattern from individual gold nanoparticles changes upon fragmentation. This passive optical readout avoids cumbersome fluorescence tagging steps.
|
|
Remarkably, by harnessing these self-assembling forces and integrating CRISPR probes within the microscopic thermally-induced flows, CRONT tweezers increase concentration and interaction rates to enable rapid single DNA detection. The team showed attomolar sensitivity for a sequence from monkeypox virus – without any target amplification. This represents up to a 10 billion-fold improvement over conventional CRISPR assays and matches PCR-level performance. Such ultrasensitive quantification on a low-cost laser tweezer system could enable point-of-care testing.
|
|
Just as notably, CRONT tweezers facilitate recognition of single nucleotide polymorphisms (SNPs) - an ability previously limited to expensive sequencing or PCR platforms. This opens possibilities for optically profiling genetic variability and mutations in situ to study biodiversity or disease mechanisms. Experimental validations showed CRONT could distinguish single base differences between SARS-CoV-2 variants.
|
|
Beyond nucleic acids, the researchers demonstrated programmable manipulation of proteins, raising intriguing prospects for studying heat-induced protein interactions. And they note the platform can be extended to diverse materials through functionalization with different probes.
|
|
While still a proof-of-concept, by combining the advantages of CRISPR assays and optothermal manipulation, CRONT tweezers overcome key barriers holding back both technologies independently. The team believes this marks an important step toward point-of-care biosensors, disease screening, remote cell manipulation, and integrating nanophotonics with programmable biological interactions. They plan to further enhance throughput to accelerate research and detection across genomics, epigenetics, and diagnostics.
|
|
If the sensitivity and simplicity can be maintained as the technology develops, CRISPR-powered optothermal tweezers could become a ubiquitous tool for laboratory and clinical analysis - bringing advanced nanotechnology to real-world biomedicine.
|

