| Apr 18, 2019 | |
Graphene foam material harvests water from air(Nanowerk News) Researchers in China have demonstrated a graphene nanocomposite foam-based water harvesting system to harvest water from air. The team reports their findings in ACS Applied Materials & Interfaces ("Superelastic Graphene Nanocomposite for High Cycle-Stability Water Capture-Release under Sunlight"). |
|
| Only 30% of all freshwater on the planet is not locked up in ice caps or glaciers (not for much longer, though). Of that, some 20% is in areas too remote for humans to access and of the remaining 80% about three-quarters comes at the wrong time and place - in monsoons and floods - and is not always captured for use by people. The remainder is less than 0.08 of 1% of the total water on the planet (read more: "Nanotechnology and water treatment") | |
| An abundant of water equivalent to about 10% of the total freshwater in lakes exists in the earth atmosphere, which can be a non-negligible freshwater resource to fight against the water shortage. | |
| That's where the graphene nanocomposite foam comes in: The foam realizes water harvesting through a capture-release cycle: | |
| 1) the capture process is composed of moisture adsorption from air by lithium chloride (LiCl) and water preservation by poly(vinyl alcohol) (PVA) and | |
| 2) the release relies on the solar-to-thermal transformer, reduced graphene oxide (rGO), to facilitate evaporation. In addition, polyimide is employed as a substrate material for the purpose of 3D porous structure formation and mechanical property enhancement. | |
 |
|
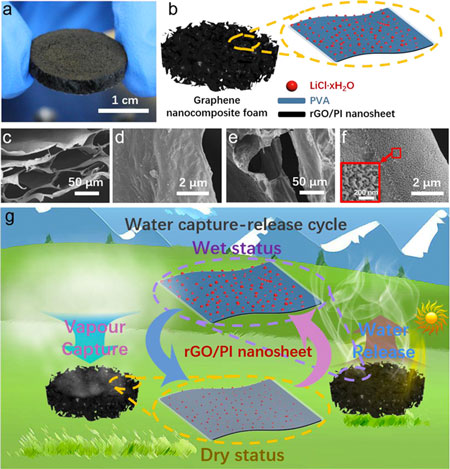
| Photograph, schematic diagram, and SEM images of the graphene nanocomposite foam. (a) Photograph of the graphene nanocomposite foam. (b) Schematic diagram of the graphene nanocomposite foam. Foam was prepared through a three-step process: freeze-drying, thermal annealing, and hydrophilic treatment. rGO/PI nanosheet, as the basic unit, can achieve the water harvesting capture-release cycle without additional energy input. (c) SEM image presents a porous structure of the rGO/PI foam without hydrophilic treatment. (d) Magnified SEM image of the rGO/PI foam without hydrophilic treatment to show a relatively smooth surface of the nanosheet. (e) SEM image of the graphene nanocomposite foam after hydrophilic treatment. (f) Magnified SEM image of the hydrophilic rGO/PI foam with bumped nanostructures. (g) Schematic diagram of the water vapor capture-release cycle. LiCl and PVA were responsible for the water capture and water storage, respectively. Adsorbed water was stored as crystallized water in LiCl hydrates and the free water molecules were restrained by hydroxyl groups on PVA through the hydrogen bond, which led to the transformation of the nanosheet from dry status to wet status. Opposite procedure, from wet status to dry status, was realized by the rGO converting the solar energy to thermal energy to facilitate water evaporation under irradiation. (Reprinted with permission by American Chemical Society) (click on image to enlarge) | |
| The as-fabricated foam can adsorb water up to 2.87 g per gram in 24 hours at a relative humidity of 90% and a temperature of 30°C, and release almost all the uptake water when it is exposed under a flux of 1 sun (1000 W per square meter, equal to the light intensity of natural sunlight) for 3 hours. | |
| At the same time, the functional foam shows superelasticity, lightweight, and remarkable reusability, thus revealing its possibility to practical use. | |
| The researchers write that, even though the rGO/PI nanocomposite foam can harvest freshwater from air, it is essential to enhance water harvesting efficiency. | |
| "Another big challenge impedes the water harvesting system utilization to explore a more cost-effective way to prepare the products," they conclude. "Though the three-step synthesis method and the composition of the foam have been optimized, it is still necessary to reduce the cost and increase the fabrication efficiency. Meanwhile, environmentally friendly materials are recommended, which would take the water harvesting system one step further to commercial application and large-scale production." |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Subscribe to a free copy of one of our daily Nanowerk Newsletter Email Digests with a compilation of all of the day's news. |
