| Posted: January 26, 2007 |
DNA gets new twist: scientists develop unique DNA nanotags |
|
(Nanowerk News) Carnegie Mellon University scientists have married bright fluorescent dye molecules with DNA nanostructure templates to make nanosized fluorescent labels that hold considerable promise for studying fundamental chemical and biochemical reactions in single molecules or cells. The work, published online Jan. 26 in The Journal of the American Chemical Society (" Fluorescent DNA Nanotags: Supramolecular Fluorescent Labels Based on Intercalating Dye Arrays Assembled on Nanostructured DNA Templates"), improves the sensitivity for fluorescence-based imaging and medical diagnostics.
|
|
"Our DNA nanotags offer unprecedented densities of fluorescent dyes and, thus, the potential for extremely bright fluorescent labels," said graduate student Andrea Benvin, who developed the nanotags in the laboratory of Bruce Armitage, associate professor of chemistry in the Mellon College of Science (MCS) at Carnegie Mellon. "We've put it all into a very small package, which will allow us to detect molecules with great sensitivity without interfering with the biological processes we are trying to understand."
|
|
The high brightness of the nanotags should be of great help in detecting rare cancer cells within tissue biopsies, for example, which is important in determining whether treatments have been successful or if recurrence is likely, according to Armitage. In addition, DNA nanotags offer the opportunity to perform multicolor experiments. This feature is extremely useful for imaging applications, Armitage says, because the multiple colors can be seen simultaneously, requiring only one experiment using one laser and one fluorescence-imaging machine.
|
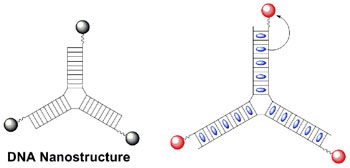 |
| (Image: Carnegie Mellon)
|
|
"For example, two different populations of cells, one healthy and the other cancerous, could be distinguished based on labeling them with different color fluorescent nanotags," Armitage said.
|
|
Benvin, Armitage and colleagues at Carnegie Mellon's Molecular Biosensor and Imaging Center modeled their DNA nanotags on the structure of phycobiliproteins. Found in certain types of algae, such as the red and blue algae in fresh and marine waters, these proteins contain multiple, fluorescent pigments that work together to absorb light energy that is then transferred to chlorophyll, where it is used for photosynthesis. The Carnegie Mellon team has mimicked this efficient light-harvesting process in the design of their DNA nanotags to create incredibly bright, fluorescent labels.
|
|
"The primary advantages of our system are the simplicity of its design combined with the ease with which the fluorescence brightness and color can be tuned," Armitage said.
|
|
To achieve greater brightness, the Carnegie Mellon team assembled well-defined nanostructured DNA templates that bind multiple fluorescent dye molecules between base pairs in the DNA helix. This arrangement keeps dyes far enough away from each other to avoid canceling out each other's fluorescence. The DNA templates can also be modified to bind to other molecules or to the surface of a cell of interest. The innovative design creates nanotags with large light-harvesting capabilities and very high light-emission (fluorescence) intensities. Because the DNA can accommodate one dye for every two base pairs, a DNA nanostructure with 30 base pairs can bind up to 15 fluorescent dye molecules. The resulting dye-DNA complexes are approximately 15 times brighter than an individual dye molecule. And they can be made even brighter by simply increasing the number of base pairs in the DNA nanostructure.
|
|
Multicolor experiments are possible because the DNA nanotags contain "light-harvesting" dyes within the DNA helix that are excited by one wavelength of light and then transfer that excitation energy to "light-emitting" dyes on the nanotag's surface (see illustration). The light-emitting dyes can fluoresce at a different color from the light-harvesting dye. For example, one type of DNA nanotag can act as an antenna that efficiently harvests blue light and transfers that light energy to another dye within the nanostructure. The second dye then emits orange, red or even infrared light. Changing the light-harvesting dyes allows even more variation in the fluorescence color, Armitage said. |
|
The nanotags are easily assembled by mixing commercially available DNA strands and fluorescent dyes. And while the work described by the Carnegie Mellon team relied on a relatively simple two-dimensional DNA nanostructure, Armitage notes that the rapidly growing field of DNA nanotechnology is generating increasingly intricate three-dimensional nanostructures that should lead to further improvements in brightness.
|

