| Sep 04, 2008 |
Nanoparticles hunt down cancer cells
|
|
(Nanowerk News) Cancer cells can be detected then destroyed using a nanostructure designed by South Korean researchers.
|
|
Bong Hyun Chung and colleagues at the Korea Research Institute of Bioscience and Biotechnology in Daejeon base their structures on hollow gold nanoparticles ("Paramagnetic gold nanostructures for dual modal bioimaging and phototherapy of cancer cells" – free access article).
|
|
The organocatalyst had been used previously by the Princeton group to activate aldehydes. But, despite activation, the aldehyde would only react with a narrow range of highly reactive partners. Adding the inorganic catalyst - a photoactive ruthenium complex - solved this problem, generating a highly reactive alkyl radical in situ that readily attacked the activated aldehyde.
|
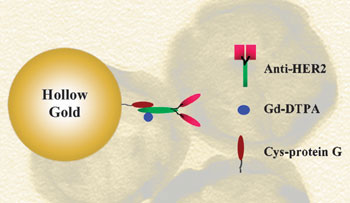 |
| The modified gold nanoparticles seek out and destroy cancer cells
|
|
The structures have antibodies on their surface which allow them to bind to cancer cells. They also contain gadolinium, which acts as a molecular resonance imaging (MRI) contrast agent and allows the cells to be seen. When Chung shone an infrared laser on the gold nanoparticles, the heat that formed destroyed the surrounding cancer cells.
|
|
The gold nanostructures overcome the drawbacks of commonly-used iron oxide MRI contrast agents, suggests Chung. Iron can lead to interference and negative contrast effects, causing errors in diagnosis. The design of the gold nanostructures leads to an enhanced signal and better diagnosis, he says.
|
|
Chung's approach is non-invasive and is likely to be effective in the treatment of early-stage cancers because it treats a specific area, unlike chemotherapy which affects the whole body. In the future 'the nanoparticles may be used for the analysis of cancer dissection in surgery', says Chung.
|
|
|

