| Aug 05, 2016 |
Making artificial cells like soap bubbles
|
|
(Nanowerk News) Researchers at the University of Tokyo used MEMS (Micro Electro Mechanical Systems) technology to produce spherical vesicles (asymmetric giant lipid vesicles/liposomes) surrounded by a membrane that, like the membrane of our cells, has an inner and outer layer composed of different phospholipids. In contrast to conventional methods of creating vesicles, this new method leaves little trace of organic solvents inside the vesicle and is an important contribution as a fundamental technique for development of artificial cell models.
|
|
Giant liposome formation methods using MEMS technology can better control the size and the encapsulated concentration compared with conventional formation methods. However, when using these formation methods based on the MEMS technology, it is impossible to avoid incorporating organic solvents in the liposome membranes, greatly affecting the stability of the liposome and rendering such methods unsuitable as a tool for researching artificial cells.
|
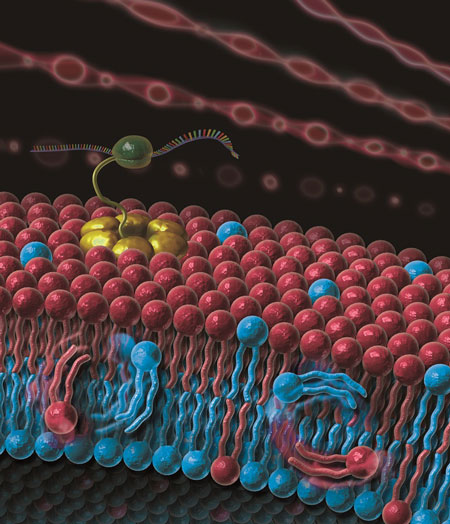 |
| A cross-sectional view of an asymmetric GV that mimics a cell membrane. The lipid flip-flop behavior and the improvement of the reconstitution ratio of the membrane proteins. This will be a useful tool for researching the mysteries of asymmetric cell membranes. (Image: Aki Sato)
|
|
Professor Shoji Takeuchi at the Institute of Industrial Science, the University of Tokyo, and Researcher Koki Kamiya at the Kanagawa Academy of Science and Technology and their colleagues have successfully produced asymmetric giant lipid vesicles (GVs) that include little organic solvent by using MEMS technology, applying a water jet to a phospholipid planar bilayer membrane in a manner similar to the creation of a soap bubble (Nature Chemistry, "Cell-sized asymmetric lipid vesicles facilitate the investigation of asymmetric membranes").
|
|
Lipids in the asymmetric GVs were observed flipping between the inner and outer layers of the membrane at approximately the same timescale as cell death occurs in living cells. In addition, when the group added peptides (protein fragments) that interact with lipid membranes, the flip-flop activity was promoted approximately tenfold compared to normal conditions. Moreover, they revealed an increase in uptake of membrane proteins when the outer layer of the asymmetric GVs contains negatively charged lipids.
|
|
Using our asymmetric GVs, we observed the dynamics of lipid molecules and the reconstitution of membrane proteins into the asymmetric lipid bilayer,” says Takeuchi. He continues, “Our asymmetric GVs provide a platform to understand the biological functions of lipid asymmetry.”
|

