| Aug 26, 2013 | |
Nanotechnology researchers increase solar cell efficiency with smart nanostructures |
|
| (Nanowerk News) Smart nanostructures can increase the yield of solar cells. An international team of researchers including physicists from the FOM Foundation, Delft University of Technology and Toyota, have now optimised the nanostructures so that the solar cell provides more electricity and loses less energy in the form of heat ("High charge-carrier mobility enables exploitation of carrier multiplication in quantum-dot films"). | |
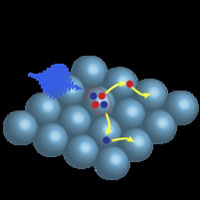 Linked quantum dots - In the new nanomaterial two or more electrons jump across the band gap as a consequence of just a single light particle (arrow with waves) being absorbed. Using special molecules the researchers have strongly linked the nanospheres (quantum dots) as a result of which the electrons can freely move and an electrical current develops in the solar cell. Solar cells A conventional solar cell contains a layer of silicon. When sunlight falls on this layer, electrons in the silicon absorb the energy of the light particles (photons). Using this energy the electrons jump across a 'band gap', as a result of which they can freely move and electricity flows. The yield of a solar cell is optimised if the photon energy is equal to the band gap of silicon. Sunlight, however, contains many photons with energies greater than the band gap. The excess energy is lost as heat, which limits the yield of a conventional solar cell. Nanospheres Several years ago the researchers from Delft University of Technology, as well as other physicists, demonstrated that the excess energy could still be put to good use. In small spheres of a semiconducting material the excess energy enables extra electrons to jump across the band gap. These nanospheres, the so-called quantum dots, have a diameter of just one ten thousandth of a human hair. Contact between quantum dots However, up until now the problem was that the electrons remained trapped in their quantum dots and so could not contribute to the current in the solar cell. That was due to the large molecules that stabilise the surface of quantum dots. These large molecules hinder the electrons jumping from one quantum dot to the next and so no current flows. |
|
| In the new design, the researchers replaced the large molecules with small molecules and filled the empty space between the quantum dots with aluminium oxide. This led to far more contact between the quantum dots allowing the electrons to move freely. | |
| Yield | |
| Using laser spectroscopy the physicists saw that a single photon indeed caused the release of several electrons in the material containing linked quantum dots. All of the electrons that jumped across the band gap moved freely around in the material. As a result of this the theoretical yield of solar cells containing such materials rises to 45%, which is more than 10% higher than a conventional solar cell. | |
| This more efficient type of solar cell is easy to produce: the structure of linked nanospheres can be applied to the solar cell as a type of layered paint. Consequently the new solar cells will not only be more efficient but also cheaper than conventional cells. | |
| The Dutch researchers now want to work with international partners to produce complete solar cells using this design. |
| Source: TU Delft |
