| Posted: Aug 05, 2014 | |
Nitrogen tunes carbon-sulfur interfaces for stable lithium-sulfur batteries |
|
| (Nanowerk Spotlight) Sulfur is a very intriguing solution for the design of high energy density storage devices. The lithium-sulfur battery theoretically delivers energy density of 2600 Wh kg-1, which is 3-5 times higher than traditional lithium-ion batteries. Unfortunately, several obstacles so far have prevented the practical demonstration of sulfur-based cathodes for Li-S batteries. Among them, the most important one is the rapid capacity fading. | |
| "The fast capacity decay of lithium-sulfur battery is ascribed to multifaceted aspects," Dr. Qiang Zhang, an associate professor at Department of Chemical Engineering at Tsinghua University, tells Nanowerk. "One of the most widely accepted reasons is assigned to the intermediate polysulfides." | |
| Polysulfides are a variety of transition forms of partially lithiated sulfur, which is highly polar and soluble in organic electrolytes. During discharge, they dissolve in the electrolyte, diffuse from cathode to anode, and react with the lithium anode. | |
| "The active materials lose in this way, undoubtedly causing capacity fading," says Zhang. "While considerable research endeavor is dedicated to solving this problem, what we are interested in is another rarely addressed issue regarding the capacity fading: the dynamic fluctuation of affinity between different sulfur species and conductive host materials." | |
| He continues to explain that, because of the multi-electron-transfer process, sulfur species vary from the initial elemental sulfur, intermediate polysulfides, and final discharge product of lithium sulfides. | |
| "Sulfur is unpolar, thus exhibits highest affinity to conventional carbon hosts," he says. "But polysulfides and lithium sulfides are highly polar, weakening the interaction between them and carbon. Due to this poor interaction, they easily detach from the carbon host and contribute no capacity. As a result, the performance of a lithium-sulfur battery deteriorates rapidly when only pure carbon hosts is employed." | |
| Consequently, he concludes, the key issue lies in how to choose an ideal host material with high affinity to both unpolar sulfur and polar polysulfides, as well as lithium sulfides. | |
| In new work published in the July 24, 2014 online edition of Advanced Materials Interfaces ("Strongly Coupled Interfaces between a Heterogeneous Carbon Host and a Sulfur-Containing Guest for Highly Stable Lithium-Sulfur Batteries: Mechanistic Insight into Capacity Degradation"), Zhang and his collaborators developed a novel strategy towards highly stable Li-S batteries by building a strongly coupled interface between surface- mediated carbon hosts and various sulfur-containing guests. | |
 |
|
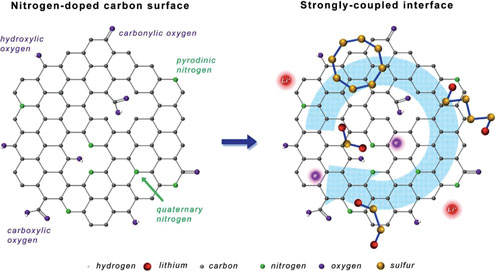
| Schematic illustration of strongly coupled interfaces between N-doped carbon host and S-containing guest for highly stable Li-S battery. (Reprinted with permission by Wiley-VCH Verlag) (click on image to enlarge) | |
| In this work, the team used nitrogen-doped carbon nanotubes as host material for the sulfur cathode: Nitrogen atoms with higher electronegativity are incorporated into the graphitic lattices of pristine carbon nanotubes, thereby providing a capability to tune their electronic structure and surface properties. | |
| How do the doping nitrogen atoms affect the electrochemical behavior when nitrogen-doped carbon nanotubes are applied to lithium-sulfur battery? | |
| Hong-Jie Peng, a graduate student in Zhang's group and the paper's first author, answers this question: "Firstly, we conducted a density functional theory (DFT) study and designed three molecular models to illustrate pure carbon, carbon with nitrogen at the edge – which we called pyridinic nitrogen – and carbon with nitrogen substituting the central carbon atom, which we called quaternary nitrogen." | |
| "Through theoretical calculations, we found that nitrogen-doped carbon nanotubes exhibited stronger interaction with polysulfides and lithium sulfides," he continues. "This is attributed to the adsorption of these polar sulfur species on the negatively charged nitrogen-doped sites. It revealed that nitrogen-doped carbon nanotubes might be worth trying as host materials." | |
| In their experiments, the team then prepared nitrogen-doped carbon nanotube/sulfur composites and assembled batteries to check if their theoretical results were reliable. | |
| "We were very happy to see that the electrochemical experiment matched our theoretical prediction very well," says Peng. "Compared to pristine carbon nanotubes-based host materials, the cycling life was significantly enhanced by six times." | |
| In conclusion, this work highlights the importance of a stable dynamic interface between carbon hosts and sulfur-containing guests and sheds new light on the lithium-sulfur battery decay mechanism. | |
| "In fact" says Zhang, "the concept of building heterogeneous cathode scaffold won?t stop here. More advanced host materials satisfying the demand of amphiphilicity to both unpolar and polar sulfur species need to be explored." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
