| Posted: Nov 11, 2016 | |
Preventing bacterial biofilm formation on catheters with nanoparticles |
|
| (Nanowerk Spotlight) In previous Nanowerk Spotlights we have reported on various therapeutic applications of nitric oxide-releasing nanoparticles: for general treatment of infections; as wound-healing agents; for the treatment of candidal burn infections; to prevent acne; even as an alternative to Viagra. | |
| In new work, researchers now have demonstrated that nitric-oxide releasing nanoparticles interfere with Staphylococcus aureus (S. aureus) adhesion and prevent biofilm formation on a rat central venous catheters model of infection. | |
| The team has reported their findings in the November 7, 2016 online edition of Antimicrobial Agents and Chemotherapy ("Sustained nitric oxide-releasing nanoparticles interfere with methicillin-resistant Staphylococcus aureus adhesion and biofilm formation in a rat central venous catheter model"). | |
| Intravenous catheters, particularly, central venous catheters (CVC), are utilized extensively in the care of patients. Unfortunately, there are over 250,000 CVC-related infections annually in the United States. According to the U.S. Center for Disease Control, between 12% and 25% of patients who acquire catheter-related bloodstream infection (CRBSI) die; many others have extended hospital stays, and increased overall treatment costs (a single incident of CRBSI can cost as much as US$ 56,000 to treat according to some studies, once the cost of pharmacy charges, catheter changes, lab tests and an additional day in the ICU are added up). | |
| S. aureus is frequently isolated in the setting of infections of CVCs and this bacterium avidly forms biofilms, which enhances bacterial resistance to medicine's current armament of antimicrobial agents and highlights the need for innovative approaches to combatting this growing threat. | |
| "Nitric oxide (NO) is a diatomic, lipophilic gaseous molecule produced by numerous cells that is involved in cellular signaling, vascular modulation and homeostasis, immune function, pro- and anti-inflammatory properties, and importantly, has bactericidal and bacteriostatic properties," Dr. Joshua D. Nosanchuk, M.D., a professor in the Departments of Medicine (Infectious Diseases) and Department of Microbiology & Immunology at the Albert Einstein College of Medicine, explains to Nanowerk. "In our new study we demonstrate that a well studied nitric oxide-releasing nanoparticle platform (NO-np) has the potential to reduce the incidence and/or treat CVC infections." | |
 |
|
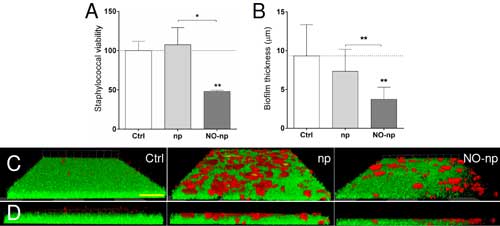
| MRSA 6498 cells within mature biofilms are effectively killed by nitric-oxide releasing nanoparticles (NO-np). Microbial biofilms were grown on polystyrene microtiter or glass-bottom plates for 24 h at 37°C and incubated in absence and presence of np or NO-np. For biofilm formation, the initial inoculum was 106 MRSA per plate. (A) The percentage viability of biofilm-associated cells was evaluated using the FDA assay. (B) The difference in biofilm thickness was examined after exposure to np or NO-np and compared with untreated control. For panels A and B, bars are the average of three wells, and error bars denote SDs. Statistical significance (*, P<0.05; **, P<0.01) was calculated by ANOVA. (C) Confocal microscopy of MRSA 6498 strain biofilms after treatment with NO-np. Images of mature bacterial biofilms showed exopolymeric matrix (red; Concanavalin A-Texas Red conjugate-stained) and bacterial cells (green; stained with SYTO9). Images were obtained after 24 h co-incubation of the bacterial cells in absence and presence of np or NO-np. (D) The thickness and morphology of the biofilms can be observed in the Z-stack reconstruction. The pictures were taken at a magnification of x100. Scale bar for all the images in C and D, 20 µm. For A-D, these experiments were performed twice, with similar results obtained each time. (© American Society for Microbiology) (click on image to enlarge) | |
| The investigators examined the formation of staphylococcal biofilms by confocal and scanning electron microscopy and found that treatment of staphylococcal biofilms with NO-np significantly reduced biofilm thickness and bacterial number compared to control biofilms. | |
| The study used in vitro models as well as a rat CVC model. When asked for comments, the collaborating senior authors Drs. Luis Martinez (New York Institutes of Technology), Josh Nosanchuk (Albert Einstein College of Medicine) and Adam Friedman (George Washington University, Deparment of Dermatology) all enthusiastically indicated that their findings present the promising nanotechnological development of NO-np for use as a prophylactic or therapeutic against bacterial biofilms on CVCs and other medical devices. | |
| The NO-np technology is currently being commercialized for a broad range of medical applications by Nano BioMed Inc. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
