| Posted: Jun 29, 2017 | |
Plasmonic nanosensor gels to detect therapeutic levels of radiation |
|
| (Nanowerk Spotlight) Ionizing radiation (e.g. X-rays, γ-rays) is widely used in the treatment of cancer, but can cause significant damage to healthy cells. The overarching goal of radiotherapy is to safely, accurately and efficiently deliver ionizing radiation in order to treat diseases, typically cancer. | |
| However, radiation-induced toxicity and accidental overexposure are serious concerns that can adversely affect patient health. That is why radiation protection of patients has emerged as an important facet of radiotherapy. Medical physicists and oncologists need to expose maximal dose to the diseased tissue while sparing surrounding tissues and organs. | |
| A novel sensor technology can help medical physicists and oncologists effectively plan fractionated radiotherapy in the clinic, reduce accidental overexposures, and reduce radiation-induced toxicity. | |
| Existing sensors used for detecting clinical doses of radiation are cumbersome, expensive, need specialized equipment and/or a highly skilled specialist for their operation. Many of these sensors cannot be molded into various shapes and size to match the contours of the body exposed to radiation. | |
| With these factors in mind, researchers in the U.S. have developed a novel gel-based device as a colorimetric sensor that can rapidly detect clinical doses of ionizing radiation. | |
| The team, led by Kaushal Rege a professor in chemical engineering at Arizona State University, Tempe, AZ in collaboration with Banner-M.D. Anderson Cancer Center in Gilbert, AZ, reports their findings in Advanced Functional Materials ("Detection of Therapeutic Levels of Ionizing Radiation Using Plasmonic Nanosensor Gels"). | |
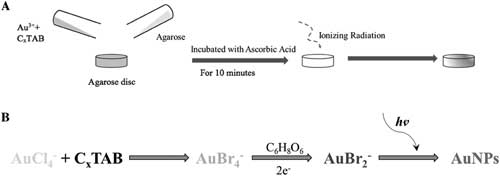 |
|
| A) Schematic of the hydrogel nanosensors for the detection of therapeutic levels of ionizing radiation (e.g., X-rays). A mixture of 10 × 10-3 m HAuCl4 and 50 × 10-3 m CxTAB (x = 12, 16) is mixed with liquid agarose and allowed to set in a circular mold to form a disc (16 mm in diameter and 3 mm in thickness). Ascorbic acid (10 × 10-3 m) is allowed to diffuse into the gel from the top for 10 min immediately prior to subjecting the disc to different doses of ionizing radiation. Irradiation of this colorless hydrogel with ionizing radiation (X-rays) results in the formation of gold nanoparticles in the hydrogel, which results in a visible change in color of the gel from colorless to maroon. B) Chemical reactions involved in the current hydrogel based nanosensor. HAuCl4 solution, which is yellow in color, changes to orange upon addition of the surfactant (CxTAB). Subsequent addition of ascorbic acid (C6H8O6) led to reduction of Au(III) to Au(I), which renders the gel colorless and translucent. Irradiation with ionizing radiation (X-rays) results in the formation of gold nanoparticles (AuNPs), which render a burgundy color to the gel due to the plasmonic properties of these nanoparticles. (Reprinted with permission by Wiley-VCH Verlag) (click on image to enlarge) | |
| "Complex fabrication procedures, high costs, cumbersome operation procedures, need for specialized equipment, need for operators with specialized training, and/or stringent environmental control (e.g. oxygen free environments) motivated us to develop a simple colorimetric sensor to detect ionizing radiation that can be translated to clinical radiotherapy applications," Rege tells Nanowerk. | |
| "The unique advantage of our sensor lies in the fact that the mode of detection is a visible change in color – typically, colorless to maroon – which makes it among the most simple detection methods available," adds Karthik Pushpavanam, the paper's first author. "This change in color is visible only 15 minutes after exposure to radiation. Increasing the amount of radiation delivered to the hydrogel increases the intensity of color developed in the gel. This change in intensity can be used for dose quantification by using a simple spectrophotometric technique." | |
| The hydrogel can be molded into various shapes depending on the user requirement. The efficacy, simplicity of operation and detection, robustness, and relative low cost make the current gel-based colorimetric sensor devices a powerful technology for detecting ionizing radiation for applications including clinical radiotherapy. | |
| These nanosensor gels can be used to report the dose of radiation delivered to different locations in the exposed area. Furthermore, this technology will help effectively plan radiotherapy treatment, which typically is administered fractions of 2 Gy dose per day leading to a cumulative dose of 20–70 Gy over the course of the treatment (1 Gy is 1 joule of energy absorbed by a mass of 1 kg). | |
| Monitoring the dose delivered during each fraction can allow for adjustments in dose as required. This, in turn, can reduce accidental overexposure and reduce radiation-induced toxicity, ultimately leading to improved patient outcomes. | |
| The team's gel-based sensor device contains gold ions in the form of a gold salt prior to ionizing radiation exposure. Ionizing radiation leads to splitting of water (hydrolysis) in the device, which results in the formation of free radicals that generate electrons. These electrons reduce gold ions to gold atoms which further nucleate and grow into gold nanoparticles. | |
| The plasmonic properties of gold nanoparticles imparts color to the originally translucent and colorless hydrogel. The development of this color is rapid and facilitates visual detection of radiation unlike current existing sensors. The intensity of the color depends on extent of nanoparticles formed in the gel, which, in turn, is reflective of the dose delivered to the gel. | |
| This intensity can be used to calibrate the gel, leading to quantitative determination of the dose delivered. | |
| The skin is at risk during all radiotherapy treatments as it is the first point of contact during radiation. Determining the absolute radiation dose entering and leaving the skin during treatment is a priority to ensure safety of the patients. | |
| In the current study, the researchers employed their hydrogel to detect and determine radiation dose delivered to the skin as well as the surface of the thorax using anthropomorphic phantoms. | |
| They were able to quantify the radiation dose with an error rate less than 10%. This sensor response was comparable to existing and commercially used sensor indicating its readiness for use in clinical applications. | |
| "We will be evaluating these gel-based detectors in animal models in order to evaluate their pre-clinical efficacy and biocompatibility," Rege describes the team's next steps. "These studies will lead to a critical evaluation of this technology before translation to actual clinical applications." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
