| Feb 04, 2019 | |
Large-scale synthesis of metal nanoclusters with thermal shocks |
|
| (Nanowerk Spotlight) Synthetically produced nanoparticles play an important role in nanotechnology. Precisely engineering the size, shape and composition of nanoparticles and nanoclusters is necessary to control their inherent electronic, optical and chemical properties, which then determine their performance in catalytic, electronic, sensing, and many other applications. | |
| Nanoclusters are small agglomerates of atoms and molecules, consisting of a few to some thousands of units and have diameters mostly in the single nanometer scale. The name nanoparticle is often used when speaking of bigger clusters with diameters from several nanometers to several hundreds of nanometers, but the distinction between a cluster and a nanoparticle is not well-defined. | |
| In a recent paper in Langmuir ("Thermal Shock Synthesis of Metal Nanoclusters within On-the-fly Graphene Particles"), researchers from the University of Maryland report significant advances in developing a high-temperature, fast nanomanufacturing technique for the large-scale production of ultra small metal nanoclusters decorated in a graphene host matrix. | |
| "Developing a fast, low-cost and scalable manufacturing technique for the large-scale production of metal nanoclusters is urgently required in order to achieve their industrial applications," Yong Yang, the paper's first author, tells Nanowerk. "In order to synthesize smaller nanoclusters with a higher number concentration, one needs to promote the nucleation rate of nanoclusters and inhibit their subsequent growth. One way to minimize the nucleation barrier is to build a high supersaturation ratio of metal atoms in the reaction volume. This can be achieved by applying a fast heat pulse to create a highly supersaturated state leading to a very fast nucleation burst." | |
 |
|
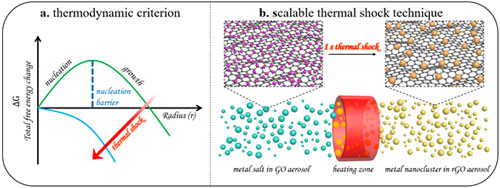
| Synthesis of metal nanoclusters via aerosol-based thermal shock technique. Left: Gibbs free energy change as a function of particle radius. Right: A schematic representation of the in-situ growth of metal nanoclusters in the one-the-fly graphene aerosol nanoreactors. (Reprinted with permission by American Chemical Society) (click on image to enlarge) | |
| In their paper, the authors report a thermal shock technique for in situ synthesis of well-dispersed metal nanoclusters in on-the-fly graphene particles. The team used the phrase on-the-fly to describe the in-motion (or flying) nature of graphene particles. | |
| To do this, they used a nebulizer to spray a precursor solution of aerosol particles containing metal salt and graphene sheets to create millions of micro-sized droplets (each about 1 micrometer in diameter) containing metal salt and graphene sheets. | |
| These droplets – which act as nanoreactors – were carried by argon gas to pass through a tubular dryer to remove the water solvent. The resulting graphene particles containing metal salt subsequently flew through a high-temperature tubular zone in which the thermal shock (about 1 second long) exerted on each graphene particle due to the thermal radiation triggered the formation of metal nanoclusters in the graphene particle while it was flying. | |
| "The size and morphology of the metal nanoclusters can be controlled by varying the reaction temperature and the mass loading of metal salt in the graphene aerosol nanoreactors," explains Yang. "Smaller nanoclusters with a narrower size distribution were obtained with a lower reaction temperature and lower mass loading." | |
| Applying their technique, the team demonstrates the formation of sub-10 nm nickel, cobalt, and tin nanoclusters in the graphene matrix. | |
| The researchers also investigated the effect of reaction temperature and metal salt loading on the formation of the nanoclusters in the graphene aerosols. | |
| "A characteristic time analysis leads to the conclusion that nanocluster formation is governed by nucleation and subsequent surface growth," notes Yang. | |
| To demonstrate a potential application, the scientists also investigated the catalytic properties of their metal nanoclusters synthesized by the aerosol-based thermal shock technique. The metal nanoclusters decorated on the graphene particles showed excellent catalytic activities towards the reduction of 4-nitrophenol by sodium borohydride. This reduction reaction has been widely used as a gold standard for investigating the catalytic properties of various nanoclusters. | |
| The team concludes that this one-step continuous aerosol-based thermal shock technique offers considerable potential for the manufacturing of well-dispersed and uniform nanoclusters stabilized within a host matrix. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
