| Jun 15, 2023 | |
Laser-induced graphenization technique advances electrofluidic paths in microfluidic paper-based devices |
|
| (Nanowerk Spotlight) Microfluidic paper-based analytical devices (µPADs) are a type of technology used for fluid analysis that leverage the properties of paper and microfluidic channels. The main advantage of these devices is their low cost, portability, and ease of use, which makes them particularly suitable for field testing, especially in resource-limited settings. They are capable of performing total (bio)chemical analysis, enhancing their utility in various fields. | |
| The operation of µPADs is based on the natural capillary action of paper, which allows fluids to be transported through small channels without the need for external pumps. This capillary action can be guided and controlled by creating hydrophobic barriers on the paper, typically through a method such as wax printing, photolithography, or plasma treatment. This allows the formation of microchannels that guide the flow of the fluid. | |
| A recent advancement in the field of µPADs is the integration of electronic components into these devices. The incorporation of electrodes, for instance, opens up new capabilities for µPADs, enabling quantitative electrochemical detection, assay miniaturization, and advanced connectivity via near-field wireless communications. However, electrode fabrication on paper presents significant challenges due to the rough and inhomogeneous nature of cellulose materials. | |
| Various techniques have been developed and refined to address these challenges, including inkjet printing, screen printing, metal sputtering, thermoplastic forming, and pencil drawing. Despite these advances, the deposited electrodes can still present limitations. They are often hydrophobic, which can hinder capillary flow, and they may display limited contact with the fluid within the paper's pores. | |
| Once the fluid is introduced into the device, it can interact with various reagents that are pre-loaded onto the paper. These reagents can then cause color changes or other observable phenomena based on the presence of specific analytes in the fluid, allowing for the detection and measurement of these analytes. | |
| Applications of µPADs span a wide range of fields, including environmental monitoring, food safety testing, and biomedical diagnostics. For example, in the field of healthcare, µPADs have been developed for the detection of a variety of diseases, including diabetes, malaria, and HIV. | |
| Even with these advances and applications, it's important to note that µPADs still face several challenges. These include the cost and complexity of fabrication techniques, the qualitative or semi-quantitative nature of their readouts, and issues with the stability and storage of reagents on the paper. However, ongoing research is aimed at overcoming these challenges and further improving the technology. | |
| In a significant breakthrough for rapid diagnostic tests, a research team at the Institute for Chemical and Bioengineering at ETH Zürich has developed an innovative technology for creating diagnostic tests with built-in electrodes. These tests enable digital result readouts, providing accurate and quantitative results through electronic signaling. | |
| The scientists report their findings in Advanced Materials ("Paper-Based Laser-Pyrolyzed Electrofluidics: An Electrochemical Platform for Capillary-Driven Diagnostic Bio assays"). | |
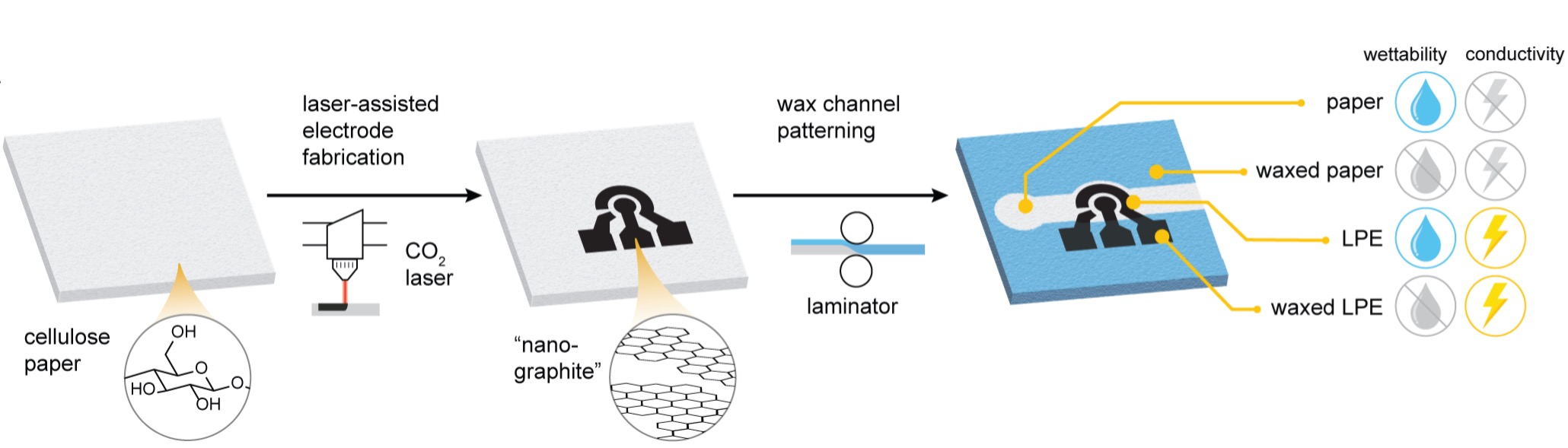 |
|
| Fabrication of paper-based electrofluidic systems. The paper embedded electrodes are created by laser-induced pyrolysis of cellulose, followed by the patterning of fluidic channels by wax lamination. This process allows for independent patterning of electrodes and channels, enabling full control over the wetting and conductive properties in different regions. (Image: Leonard Bezinge, ETH Zurich) | |
| "This platform has the potential to improve the diagnostic accuracy of existing test assays or enable the development of new test functionalities that are currently not feasible with technologies relying on colored test lines," Leonard Bezinge, a final-year PhD student at ETH Zürich, and first author of this work, tells Nanowerk. | |
| The core of the team's findings lies in a new method for integrating electrodes into paper-based test strips. They achieved this through a process called laser-induced pyrolysis of cellulose paper, which transforms the cellulose molecules into a conductive graphene-like material. The electrodes formed in this manner retain the porous and fibrous nature of the paper, crucial for capillary-driven assays like rapid diagnostic tests. | |
| "The seamless integration of electronic components into paper-based microfluidics enables the application of advanced measurement techniques in a simple and cost-effective platform," comments Bezinge. "It advances the field by improving the speed, sensitivity, and accuracy of rapid tests, facilitating reliable and efficient chemical, biological, and medical analyses." | |
| This development offers a novel approach to integrating electronics into paper, opening up new possibilities when combined with capillarity-driven microfluidics. Previous methods of electrode fabrication involved coating the top surface of the paper with a conductive ink or metal, resulting in poor integration within the cellulose paper network and negatively impacting detection efficiencies. | |
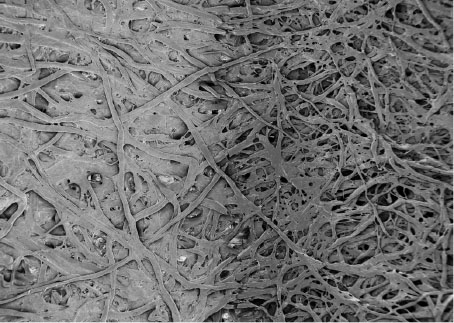 |
|
| Top-view SEM image of a cellulose-laser induced graphenic electrodes interface revealing the morphological transition. Left: cellulose; Right: laser-pyrolyzed electrodes. (Image: Leonard Bezinge, ETH Zurich) | |
| "Our motivation stemmed from the desire to combine the simplicity of existing rapid diagnostic tests with the advantages of electrochemical signaling and digital readouts," Bezinge notes. | |
| The technology provides a solution for integrating electrodes into paper-based microfluidics, enabling the combination of certain types of assays, such as flow-through devices, with electrochemical readouts. As the researchers point out, this achievement is especially significant because certain types of assays previously could not be combined with electrochemical readouts. | |
| The team's work has primarily focused on applications in medical diagnostics. They envision this technology being utilized as a rapid test technology for at-home testing of nucleic acid, antigen or antibody biomarkers, with the results read digitally from a smartphone. Furthermore, they see potential for this platform to be used for high-throughput analysis of multiple samples on a single device, where the test strip could be integrated as a single-use cartridge in a portable analyzer. | |
| The general nature and affordability of this fabrication platform anticipate a wide range of applications where quantitative electrochemical readout is required in conjunction with inexpensive single-use test strips. Bezinge predicts, "We expect to see novel approaches to building and integrating electrodes into complex three-dimensional fluidic architectures for sample processing and detection in the fields of medical diagnostics and health monitoring, environmental testing and food analysis." | |
| Léonard Bezinge explains the research results. | |
| Going forward, the researchers aim to expand the potential field of applications within disease diagnostics and explore new labeling chemistry and detection pathways that can be combined with these electrodes. | |
| "The major challenge ahead is the development of true sample-to-answer devices that integrate all of these steps into a single, user-friendly device," Bezinge concludes. "This endeavor is highly challenging and requires a multidisciplinary approach. We encourage researchers from different fields to share their design and construction files, particularly as we transition towards digital fabrication tools, to facilitate collaboration and further advancements in the field." | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
