| Nov 03, 2023 | |
Advanced liquid templating technique enables tailored aerogel synthesis |
|
| (Nanowerk Spotlight) Aerogels are a unique class of extremely porous solid materials with densities and thermal conductivities lower than air. Their impressive properties, including low density, high porosity, and low thermal conductivity, make aerogels promising for a wide range of applications. These uses include insulating spacecraft, absorbing oil spills, and shielding electronics from electromagnetic interference. | |
| Despite their potential, aerogels' widespread adoption has been hindered by their fragile nature. Aerogels are typically fabricated by extracting the liquid component from a gel. This leaves behind a matrix composed almost entirely of air with a minimal solid nanostructured network. While this generates materials with densities as low as 3 milligrams per cubic centimeter, it also results in poor mechanical properties and brittleness. Furthermore, it is difficult to engineer specific aerogel compositions and porosity levels to optimize properties for different applications. | |
| Aerogels were first invented in the 1930s by replacing the liquid in a gel with air while preserving the gel's intricate solid scaffolding. This is typically done using a specialized freeze-drying process known as lyophilization. First, the gel is frozen, which solidifies the liquid component. The frozen gel is then placed under vacuum conditions. This causes the frozen liquid to change directly from a solid to a gas, through a process known as sublimation. What remains is just the porous solid network that previously encapsulated the liquid. | |
| The result is a solid foam composed of over 90% air with densities rivaling the lowest theoretically achievable. However, the minimal solid framework makes aerogels extremely brittle and fragile. A larger solid fraction would improve durability, but also increase weight and compromise their uniquely low densities and thermal conductivities. This fragility has hampered their real-world deployment, despite their almost magical properties. | |
| To improve the gels’ durability while retaining their light weight, researchers at the University of British Columbia devised a way to template their structure using two immiscible liquids. They report their findings in Advanced Materials ("Liquid-Templating Aerogels"). | |
 |
|
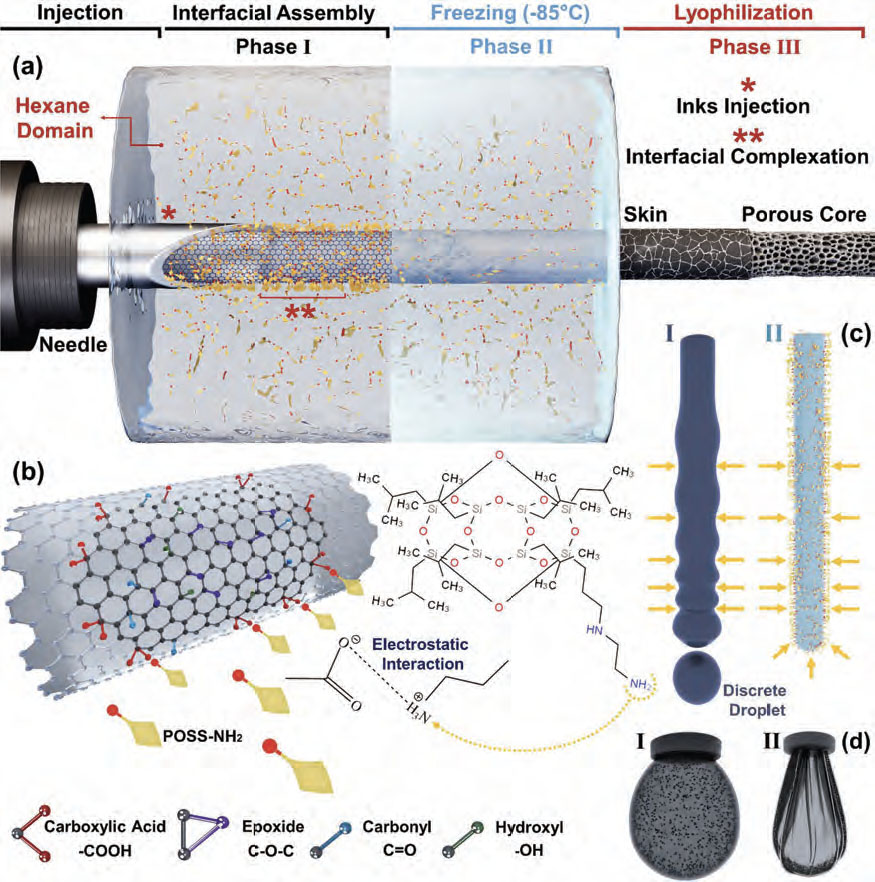
| Schematic illustration of the liquid templating methodology. a) The coined process for generating hierarchical 3D aerogels with desired components and functionality, starting from injecting aqueous phase containing GO into an immiscible liquid, i.e., hexane, containing POSS-NH2 (Phase I) to freezing of liquid template (Phase II) and subsequent lyophilization to generate filamentous aerogels (Phase III). The aerogels comprise core–shell filaments with a thin solid skin wrapped around a porous core. In Phase I, * showcases the injection of aqueous GO-based inks into the hexane domain containing POSS-NH2, and ** indicates the interfacial complexation, viz., electrostatic interaction of ligands and NPs. As illustrated in Phase II, the placement of the aqueous liquid template into a freezer at −85 °C leads to its freezing, whereas the hexane domain remains liquid owing to its low freezing point (−95.3 °C), enabling its separation before lyophilization. b) Electrostatic interaction of POSS-NH2 molecule with GO through carboxylic acid deprotonation and amine functional group protonation. c) I) Break up of the liquid stream into discrete droplets to suppress instabilities, and II) enhanced integrity of the liquid stream upon formation of NPSs at the interface, locking in the non-equilibrium shape of the liquid construct in the form of the filament. d) The shape of the droplet in pendant drop tensiometry, I) in equilibrium without the presence of POSS in the oil phase and II) in non-equilibrium shape upon the formation of a solid skin around the droplet as a result NPSs jamming, holding the integrity of liquid construct; the black dots in the image represent the GO dispersed in the aqueous phase. (Reprinted with permission from Wiley-VCH Verlag) | |
| The team starts by injecting an aqueous solution containing nanoparticles, like graphene oxide or cellulose nanofibers, into a nonpolar liquid such as hexane. The nanoparticles rapidly migrate to the interface between the liquids and bind to complementary molecules pre-dissolved in the hexane. This forms a dense layer encapsulating the aqueous solution within the non-polar liquid. Freezing and then freeze-drying this assembly leaves behind an aerogel with a solid outer skin enveloping a highly porous inner network. Varying the nanoparticles and other reaction conditions allows precise control over the aerogel’s composition and multiscale porosity. | |
| The researchers leveraged this tunability to optimize aerogels for blocking electromagnetic interference (EMI). With fine-tuned properties, the materials achieved EMI shielding effectiveness rivaling metal foils, while weighing mere milligrams per cubic centimeter. This light weight makes them promising for aerospace and aviation applications where added mass from shielding is highly detrimental. The gels also rapidly absorb oils up to 487 times their own weight. | |
| Lead author Milad Kamkar explains the significance of the developments: “Liquid templating through interfacial complexation is a strategy that can revolutionize the design of materials, offering a promising approach to enhancing performance far beyond what can be achieved today.” | |
 |
|
| Production of aerogels with different compositions and characteristics, including: I) GO aerogel, II) GO-Fe aerogel, and III) GO-CNF aerogel; in this image, the brownish flakes in (II) represent the GO-Fe3O4 flakes and the fibers on the aerogel in (III) denote CNFs. (Reprinted with permission from Wiley-VCH Verlag) | |
| The new aerogels address two persistent challenges hampering real-world uses of these solid foams. First, their extreme fragility, which often limits applications due to poor mechanical stability. And second, the difficulty of pre-defining aerogel properties by engineering their composition and porosity. This work overcame both issues through judicious choice of nanoparticle building blocks and precise structuring of the gels by liquid templating. | |
| The result is ultra-lightweight aerogels around 3 milligrams per cubic centimeter with compression resilience up to 90% strain - among the highest reported. Furthermore, selecting the aqueous phase nanoparticles provides in-situ control over the aerogels’ chemical makeup. This allowed tailoring them for either exceptional EMI attenuation or oil absorption simply by using graphene oxide versus graphene and iron oxide nanoparticles. | |
 |
|
| Non-reduced and reduced GO aerogels on the very light hairs of cactus. (Reprinted with permission from Wiley-VCH Verlag) | |
| While aerogels have been around for decades, their brittleness and lack of tunability have constrained real-world applications. By fabricating the gels around templating liquids, the researchers achieved exquisite control over the materials’ composition and porosity. Their processing advancements pave the way for durable, customizable aerogels that solve pressing needs for electromagnetic shielding and oil spill remediation. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
