| Oct 28, 2022 |
MOF and graphene-based nanoconfinement strategy enhances oxygen evolution performances
(Nanowerk News) Prof. ZHANG Tao's group at the Ningbo Institute of Materials Technology and Engineering (NIMTE) of the Chinese Academy of Sciences (CAS), in collaboration with Prof. HOU Yang from Zhejiang University and Prof. XIAO Jianping from the Dalian Institute of Chemical Physics of CAS, proposed a novel two-dimensional (2D) nanoconfinement strategy to strongly enhance the oxygen evolution reaction (OER) activity of low-conductivity metal-organic frameworks (MOFs).
|
|
Results were published in Nature Communications ("Exceptional catalytic activity of oxygen evolution reaction via two-dimensional graphene multilayer confined metal-organic frameworks").
|
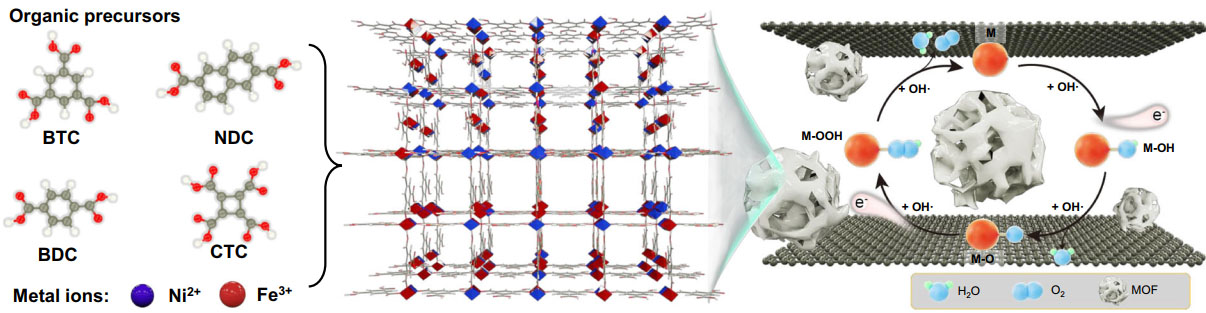 |
| The as-prepared NiFe-MOF//G via the nanoconfinement from graphene multilayers. (Image: NIMTE)
|
|
The development of high-efficiency electrocatalysts for the electrochemical conversion of water to generate environmentally friendly and sustainable hydrogen energy has drawn tremendous attention for decades.
|
|
Despite the crucial role the OER plays in water splitting, OER at the anode eagers for a relatively high thermodynamic potential to accelerate water splitting kinetics. Thanks to the large surface area, tunable porosity, diverse compositions and metal centers, MOFs have emerged as promising candidates for efficient OER electrocatalysts. However, the intrinsic poor conductivity of the most MOFs seriously impeded their catalytic activity.
|
|
To address this issue, researchers at NIMTE proposed an electrochemical strategy to confine MOFs between graphene multilayers via the two-electrode electrochemical system, thus endowing poorly conductive MOFs with strongly enhanced catalytic performance.
|
|
The as-prepared NiFe-MOF//G shows a remarkably low overpotential of 106 mV to reach 10 mA cm-2, surpassing the pristine NiFe-MOF as well as other previously reported MOFs and their derivatives. Besides, the NiFe-MOF//G electrode is highly stable, which can retain the performance for more than 150 h at 10 mA cm-2 without obvious activity decay.
|
|
Notably, the results of X-ray absorption spectroscopy experiments and density-functional theory calculations indicate that the nanoconfinement from graphene multilayers optimizes the electronic structure and catalysis center of MOF materials with the formation of highly reactive NiO6-FeO5 distorted octahedral species in MOF structure. In addition, the nanoconfinement lowers limiting potential for water oxidation reaction.
|
|
The nanoconfinement strategy can be applied to other varied MOFs with different structures, greatly improving their electrocatalytic activities. Meanwhile, this work challenges the common conception of pristine MOFs as inert catalysts, and reveals the great application potential of poorly conductive or even insulating MOFs into electrocatalysis applications.
|

