| Oct 22, 2012 |
The nanomechanical signature of breast cancer
|
|
(Nanowerk News) The spread of cancer cells from primary tumors to other parts of the body remains the leading cause of cancer-related deaths. The research groups of Roderick Lim and Cora-Ann Schoenenberger from the Biozentrum of the University of Basel, reveal in the journal Nature Nanotechnology ("The nanomechanical signature of breast cancer") how the unique nanomechanical properties of breast cancer cells are fundamental to the process of metastasis. The discovery of specific breast cancer “fingerprints” was made using breakthrough nanotechnology known as ARTIDIS. Lim’s team has now been awarded about 1.2 million Swiss francs from the Commission for Technology and Innovation (CTI) to further develop ARTIDIS.
|
|
Breast cancer is the most common form of cancer in women with 5500 patients being diagnosed with the disease in Switzerland each year. Despite major scientific advancements in our understanding of the disease, breast cancer diagnostics remains slow and subjective. Here, the real danger lies in the lack of knowing whether metastasis, the spread of cancer, has already occurred. Nevertheless, important clues may be hidden in how metastasis is linked to specific structural alterations in both cancer cells and the surrounding extracellular matrix. This forms the motivation behind ARTIDIS (“Automated and Reliable Tissue Diagnostics”), which was conceived by Dr. med. Marko Loparic, Dr. Marija Plodinec and Prof. Roderick Lim to measure the local nanomechanical properties of tissue biopsies.
|
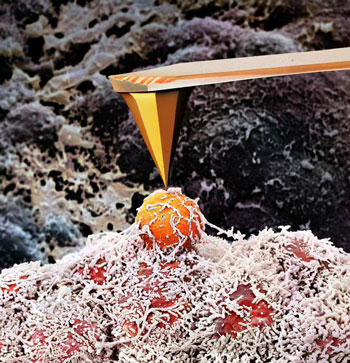 |
| Using ARTIDIS to feel the tissue structure of a tumor biopsy by a nanometer-sized atomic force microscope tip. (Image: Martin Oeggerli)
|
|
“Fingerprinting” breast tumors
|
|
At the heart of ARTIDIS lies an ultra-sharp atomic force microscope tip of several nanometers in size that is used as a local mechanical probe to “feel” the cells and extracellular structures within a tumor biopsy. In this way, a nanomechanical “fingerprint” of the tissue is obtained by systematically acquiring tens of thousands of force measurements over an entire biopsy. Subsequent analysis of over one hundred patient biopsies could confirm that the fingerprint of malignant breast tumors is markedly different as compared to healthy tissue and benign tumors. This was validated by histological analyses carried out by clinicians at the University Hospital Basel, which showed a complete agreement with ARTIDIS. Moreover, the same nanomechanical fingerprints were found in animal studies initiated at the Friedrich Miescher Institute.
|
|
Plodinec, first author of the study, explains: “This unique fingerprint reflects the heterogeneous make-up of malignant tissue whereas healthy tissue and benign tumors are more homogenous.” Strikingly, malignant tissue also featured a marked predominance of “soft” regions that is a characteristic of cancer cells and the altered microenvironment at the tumor core. The significance of these findings lies in reconciling the notion that soft cancer cells can more easily deform and “squeeze” through their surroundings. Indeed, the presence of the same type of “soft” phenotype in secondary lung tumors of mice reinforces the close correlation between the physical properties of cancer cells and their metastatic potential.
|
|
ARTIDIS in the clinics
|
|
“Resolving such basic scientific aspects of cancer further underscores the use of nanomechanical fingerprints as quantitative markers for nanotechnology cancer diagnostics with the potential to prognose metastasis.”, states Loparic, who is project manager for ARTIDIS. On an important practical note, a complete biopsy analysis by ARTIDIS currently takes four hours in comparison to conventional diagnostics, which can take one week. Based on the potential societal impact of ARTIDIS to revolutionize breast cancer diagnostics, Lim’s team and the Swiss company Nanosurf AG have now been awarded about 1.2 million Swiss francs by the Commission for Technology and Innovation (CTI) to further develop ARTIDIS into a state-of-the-art device for disease diagnostics with further applications in nanomedicine.
|
|
Over the next two years, Lim and colleagues will engage and work closely with clinicians to develop ARTIDIS into an easy-to-use “push-button” application to fingerprint diseases across a wide range of biological tissues. As a historical starting point, the first ARTIDIS demo-lab has already been established at the University Hospital Eye Clinic to collect data on retinal diseases with the goal of improving treatment strategies.
|

