| Jan 22, 2019 | |
From atoms to nanoparticles to living matter |
|
| (Nanowerk Spotlight) Atoms form molecules or crystals using their outermost electrons through interactions between single elements or multiple elements. Various molecules and crystals can assemble in different ways to form organic structures, such as DNA and cell-membrane phospholipids, or various tissues. These tissues are the bases of organs in a living organism. | |
| Whereas the evolutionary path of the atomic world has occurred over billions of years, the evolutionary path of the synthetic nanoparticle world has just begun. Man-made nanoparticle assemblies are beginning to revolutionize different fields including thermoelectronics, photoelectronics, catalysts, energy generation and storage, as well as medical diagnostics and therapeutics. | |
| A review article in Advanced Functional Materials ("From Atoms to Lives: The Evolution of Nanoparticle Assemblies") uses the unconventional approach of comparing the atomic and nanoparticle worlds to describe the development of nanoparticle research during the past several decades and provide pointers as to what the future might hold. | |
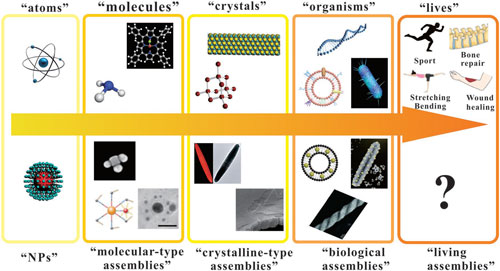
| The authors discuss in great detail corresponding classifications, such as atoms and nanoparticles; molecules and molecular type nanoparticle assemblies; crystals and crystalline-type nanoparticle assemblies; biological organisms and analogous artificial assemblies; and biologically functional nanoparticle complex assemblies. | |
 |
|
| The evolutionary path of the atomic world and architectural evolution of nanoparticle world. (Reprinted with permission by Wiley-VCH Verlag) (click on image to enlarge) | |
Similarities and differences between atoms and nanoparticles |
|
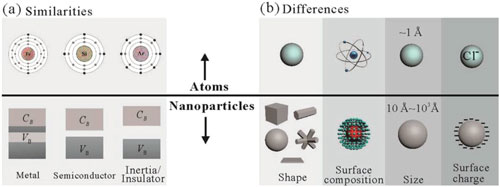
| The configuration of nanoparticles is similar to that of atoms. An atom has a nucleus and surrounding electron clouds, while an nanoparticle consists of a core element and coating ligands. The electrons balance the positive charge of nuclei and are responsible for interactions with other atoms. The ligands of nanoparticles maintain the stability of the core materials, render them soluble in certain solvents, and enable their interactions with target objects. | |
| Atoms can gain or lose electrons to become ions. Modifying the coating ligands of nanoparticles can change the surfaces of the nanoparticles to become positively or negatively charged or even enable the formation of crystalline or defective structures. | |
| Atoms can be categorized into different groups with specific properties, such as metals, semiconductors, and inert elements, depending on their numbers of protons and electrons. nanoparticles can similarly be categorized into metallic, semiconductor, and insulator types according to the band theory of the nanoparticle cores. | |
| However, nanoparticles are unlike atoms in that their physical and chemical properties, such as size, surface composition, surface charges, flexibility, and shapes govern the functionalities of nanoparticles in various applications. Generally, The advantage of using nanoparticles as building blocks instead of atoms is their complex and tunable configurations and functions. | |
| Usually, the center of the atomic nucleus is considered as hard core and the boundary of the surrounding cloud of electrons can have flexibility to engender polarization or deformation in ionized and covalent bonding states. In contrast, nanoparticles show more flexibility and coordinability which manifest for independent existence at states, deformation controllability, flowability and designability. | |
 |
|
| a) Similarities and b) differences between atoms and nanoparticles. (Reprinted with permission by Wiley-VCH Verlag) (click on image to enlarge) | |
| The essential characterization of nanoparticles is the adjustable size at least one direction of less than 100 nm. While the atomic size usually depend on the state and context of the boundary of the surrounding cloud of electrons, and its size vary in a predictable and regular manner across the periodic table. In terms of surface composition, the periphery of an atomic nucleus has only electrons. | |
| However, the surface of nanoparticles can accept coatings of ligands or shells to form interfacial layers including metal/inorganic shells, polymetallic salts, organic chains, functionalized derivatives, polymers, cell membranes, and even biofriendly DNA sequences. | |
| Geometrically, atoms can be modeled as spherical entities while nanoparticles can have various shapes and structures, i.e., spherical and nonspherical. Nonspherical nanoparticles (cubes, plates, rods) play a key role in the nanoparticle field because they can lead to specific superstructures and functions. | |
| Core?shell nanoparticles is another important group of nanoparticles that differs from atoms. A wide range of different materials and combinations can be used as their constituent components, and the choice of the core or shell materials is strongly dependent on the end use. | |
Molecules and molecular-type assemblies |
|
| The world is not built from individual atoms or nanoparticles – it is their assemblies that form our world and the novel nanoworld. | |
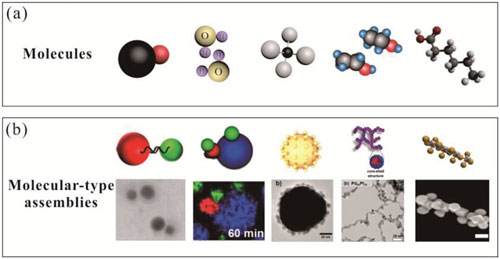
| Atoms bond in different ways to form molecules where they share or exchange electron density. Atoms can form small inert molecules and optically, thermally, or functionally responsive molecules and macromolecules as well as natural biopolymers such as proteins that are fundamental to biological structure and function. | |
| Extending the comparison of atoms to nanoparticles, the authors suggest that nanoparticles with non-oriented interparticle interactions can be regarded as molecular-type assemblies. This definition provides a simple way to broadly classify nanoparticles forming molecular-type assemblies. | |
| Here, the formed molecular-type nanoparticle assemblies are actually the unregular assembly of nanoparticles. The random aggregation among nanoparticles occur in an arbitrary activated site similar to the construction of amorphous atomic materials, such as amorphous metals, and glasses, with disordered atomic-scale structure. | |
 |
|
| a) Different types of molecules. b) Corresponding molecular-type assemblies. (Reprinted with permission by Wiley-VCH Verlag) (click on image to enlarge) | |
Crystals and crystalline-type assemblies |
|
| Compared with molecules, crystals have long periodical and orientational symmetry. Generally, the symmetry in crystals results from periodical and orientational arrangements of monoatomic/ polyatomic interactions via ionic, covalent, or metallic bonds. | |
| Nanoparticle superlattices have similar characteristics in terms of their structure and interparticle interactions. nanoparticles with oriented interparticle interactions can be classified as crystalline-type assemblies. Under this framework, single/binary/multicomponent nanoparticle superlattices correspond to monoatomic/ diatomic/ polyatomic crystals. | |
| Compared to the interaction bonds between atoms in crystals, the interparticle interaction forces of crystalline-type nanoparticle assemblies tend to bond weakly through noncovalent interaction, such as van der Waals interactions, electrostatic interactions, and hydrogen bonds. | |
| A key feature of this type of nanoparticle assembly is that the interparticle interactions are isotropic and cannot be individually modified. As demonstrated in crystalline-type nanoparticle assemblies, interparticle interactions can now be tuned to provide orientational and translational properties. | |
Biological organisms and analogous artificial assemblies |
|
| Prebiotic structures of amines, amino acids, thiols, and other molecules in Earth?s primordial era facilitated the formation of biological assemblies such as phospholipids and DNA. These molecules combined chemically to form the essential part or basic structure of organelles and tissue structures. There is some indication that biological organisms played an important role in primordial Earth conditions to facilitate living organism progress. | |
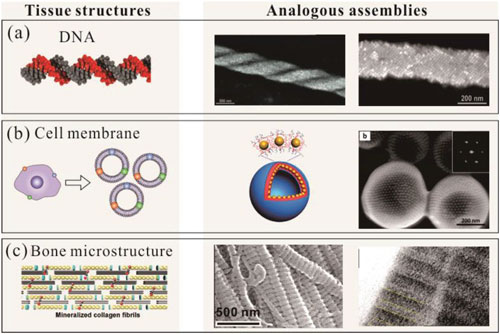
| The authors argue that biological-like artificial assemblies such as DNA, the phospholipid layers of cell membranes, and the hierarchical structures of living organisms can be associated with the functional properties of nanoparticles present in a wide variety of catalytic, optical, and biomedical processes. | |
 |
|
| Different types of biological tissue structures and analogous nanoparticle assemblies. a) The chirality screw structures of DNA. b) The phospholipid layer structures of the cell membrane. c) The hierarchical structures of bone tissues. (Reprinted with permission by Wiley-VCH Verlag) (click on image to enlarge) | |
| The fusion of inorganic nanoparticles with living matter can provide new systems that combine the advantages of both worlds. For instance, researchers have fabricated self-photosensitizing nonphotosynthetic bacteria by hybridizing inorganic semiconductors so that they could efficiently harvest sunlight with biocatalysts specializing in energy transformation ("Self-photosensitization of nonphotosynthetic bacteria for solar-to-chemical production"). | |
| In this work, the researchers grew the nonphotosynthetic CO2-reducing bacterium Moorella thermoacetica and bioprecipitated it with CdS nanoparticles, transferring CO2 into acetic acid via photosynthesis. This Moorella thermoacetica?CdS system had better performance and greater potential applications due to its ∼90% photosynthetic transfer rate, nonenergy loss during the dark periods, and tunable light flux. | |
| Concluding their review, the authors point out that current nanoparticle assemblies are static, i.e., they cannot self-respond/adapt to changing environments. | |
| "Beyond dynamic nanoparticle assemblies, an ambitious target for nanoparticle assembly research is to produce, or create, living nanoparticle assemblies. Certain experiments have already been performed. nanoparticle-assisted biosystems, bioassisted nanoparticle systems, and bio/nanoparticle coassistance systems are gradually becoming more prominent, suggesting a totally different future for nanoparticle assemblies." | |
| Three categories of functional assemblies, that are also the basic features of living organism – for motion, for self-repair, and for self-regulation/ adaptation – will drive the future of nanoparticle assemblies and open the door to an era where DNA-based living nanoscale materials are 'genetically' modifiable and can undergo structural 'evolution'. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
