| Jul 03, 2020 | |
The superiority of single-layer molybdenum disulfide for water desalination |
|
| (Nanowerk Spotlight) Most available desalination technologies rely on a process called reverse osmosis (RO) to push seawater through a membrane in order to produce fresh water. This process has always been plagued by high energy usage and fouling issues. | |
| The membranes used in RO have tiny holes in them to keep back salt and other impurities. Although thin to the eye, these membranes are still relatively thick for filtering on the molecular level, so a lot of pressure has to be applied to push the water through. | |
| In the search for more efficient alternatives, scientists are intensely researching atomically thin two-dimensional (2D) materials and metal-organic frameworks in order to find better substitutes for traditional water desalination membranes (read more: "Metal-organic frameworks as high-performance water desalination membranes"). | |
| For instance, researchers have demonstrated that nanofluidic membranes based on pristine and chemically modified graphene oxide membranes effectively block organic dyes and nanoparticles as small as 9 Ångström, i.e. 0.9 nm (read more: "Novel desalination method with nearly perfect salt rejection and high water flux"). | |
| In new work, reported in ACS Energy Letters ("Why is Single-Layer MoS2 a More Energy Efficient Membrane for Water Desalination?"), researchers now have shown with molecular dynamics simulations that a nanoporous MoS2 membrane allows a higher water flux compared with other materials such as graphene, boron nitride and phosphorene. | |
 |
|
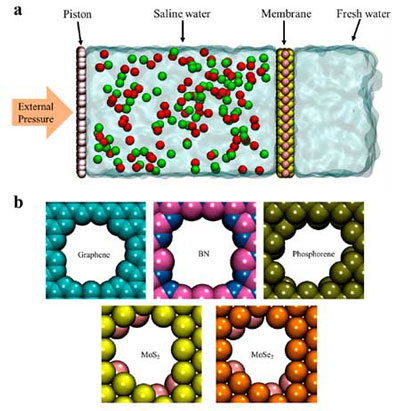
| (a) Visualization of a typical reverse osmosis water desalination simulation for 2D membranes: a graphene piston, saline water section with ions, membrane with nanoscale pore (MoS2 in this schematic), and filtered freshwater section behind the membrane. (b) Snapshots of ∼50 Å2 nanopores drilled in five different membrane materials. (Reprinted with permission by American Chemical Society) | |
| Recent experiments have already shown the viability of laminar MoS2 in practical water desalination applications ("2D nanomaterial reverse osmosis membrane for water desalination"). This new finding indicates that MoS2 can be a much more energy-efficient candidate for water desalination and a potential solution for people living in freshwater-scarce regions of the world. | |
| "In this work, we dug deeply into the physical reasons behind why MoS2 performed better than other two-dimensional materials in water desalination processes," Amir Barati Farimani, an Assistant Professor of Mechanical Engineering at Carnegie Mellon University, tells Nanowerk. "Through molecular dynamics simulation, we found out that water density near a MoS2 membrane is lower compared to that near other materials, which indicates that water molecules were more likely to transport through the membrane instead of accumulating around it." | |
| Farimani's team also discovered that a MoS2 pore, even with the same area as graphene and other 2D materials' pores, generally held more water inside of it and allowed faster transport of water molecules. Most importantly, the scientists calculated that the energy barrier for water molecules to pass MoS2 pores was lower compared with other materials. | |
| These reasons combined lead to higher performance of MoS2 membranes in water desalination processes. | |
| To fairly compare the performance of MoS2, graphene, phosphorene, boron nitride (BN), and molybdenum diselenide (MoSe2) in water desalination, the team designed the pore sizes of all four materials the same. | |
 |
|
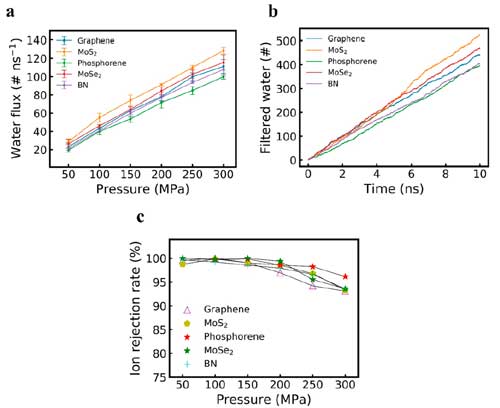
| (a) Water flux of graphene, MoS2, phosphorene MoSe2, and BN with the same size pore, with respect to applied external pressure. Error bar on each data point represents one standard deviation. (b) Number of filtered water molecules with respect to simulation time for different porous material membranes under 100 MPa external pressure. (c) Ion rejection rate of graphene, MoS2, phosphorene, MoSe2, and BN with respect to the applied external pressure ranging from 50 to 300 MPa. (Reprinted with permission by American Chemical Society) (click on image to enlarge) | |
| "We found the water flux of the MoS2 membrane the highest," says Farimani. "With 100 MPa pressure applied to saline water, the average water flux is 55.17 #/ns, 40.01 #/ns, 43.27 #/ns, 40.93 #/ns, and 46.09 #/ns for MoS2, phosphorene, graphene, BN, and MoSe2, respectively. These molecular dynamics simulation results support the assumption that nanoporous MoS2 membranes, if applied in real-world large scale water desalination, can greatly reduce energy consumption." | |
| According to the team, the future directions for this research will be to experimentally realize a water desalination process using MoS2 membranes with nanopores. This could also open new opportunities for more applications of MoS2 in areas such as supercapacitance. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
