| Apr 12, 2022 | |
How advanced nanotechnology drives ultrathin membrane development for separations |
|
| (Nanowerk Spotlight) Ultrathin membranes play an important role in the desalination process and can transform even the dirtiest water into something potable for millions of people across the world (read more about the role of nanotechnology in water treatment). | |
| Separation and purification of bio-molecules such as proteins and viruses are also important processes in the biotechnology and pharmaceutical industries. The risk of contamination with either known or unknown viruses in biological or therapeutic products require production processes that completely eliminate the risks of virus contamination. | |
| Driven by advanced nanomaterials and nanofabrication technologies, tremendous progress has been made over the last 15 years in making ultrathin membranes based on a variety of advanced nanomaterials and nanotechnologies. | |
| Depending on the type of materials, membranes of different thicknesses have been termed as 'ultrathin' in literature, ranging from atomic thickness to a few micrometers, although most works focus on thicknesses below 100 nm. | |
| Further pushing membrane thickness to sub-50 nm or even atomic level is appealing to researchers, with the ultrafast permeance that may be enabled and ultimate understanding of formation and transport at the extremely low thickness that can be significantly different from their thicker counterparts. | |
| The fabrication of 15 nm-thin silicon membranes in 2007 marks the beginning of this new era, where a porous silicon membrane was fabricated through silicon deposition, annealing, and etching (Nature, "Silicon for the perfect membrane"). | |
| As a new review article in Advanced Materials ("Ultrathin Membranes for Separations: A New Era Driven by Advanced Nanotechnology") points out, the decisive problem is how to minimize the thickness of selective layers while maintaining high separation accuracy and sufficient mechanical stability. | |
| Solutions to this challenge require: 1) facile and efficient fabrication methods that are preferably capable of precisely controlling the thickness and avoiding nonselective defects; 2) mechanically robust selective layers and 3) proper support layers. | |
| In addition, in view of practical applications and commercialization, uniform pore size, high porosity, adequate stability, and acceptable cost are also important factors to be taken into account. Advanced materials and technologies are thus keys to achieving sub-50 nm thickness for membrane separations. | |
| In their review, the authors provide a thorough review of sub-50 nm membranes, from fabrication methods, membrane structures to applications. For this purpose, they classify membranes into network, laminar, and framework structures. | |
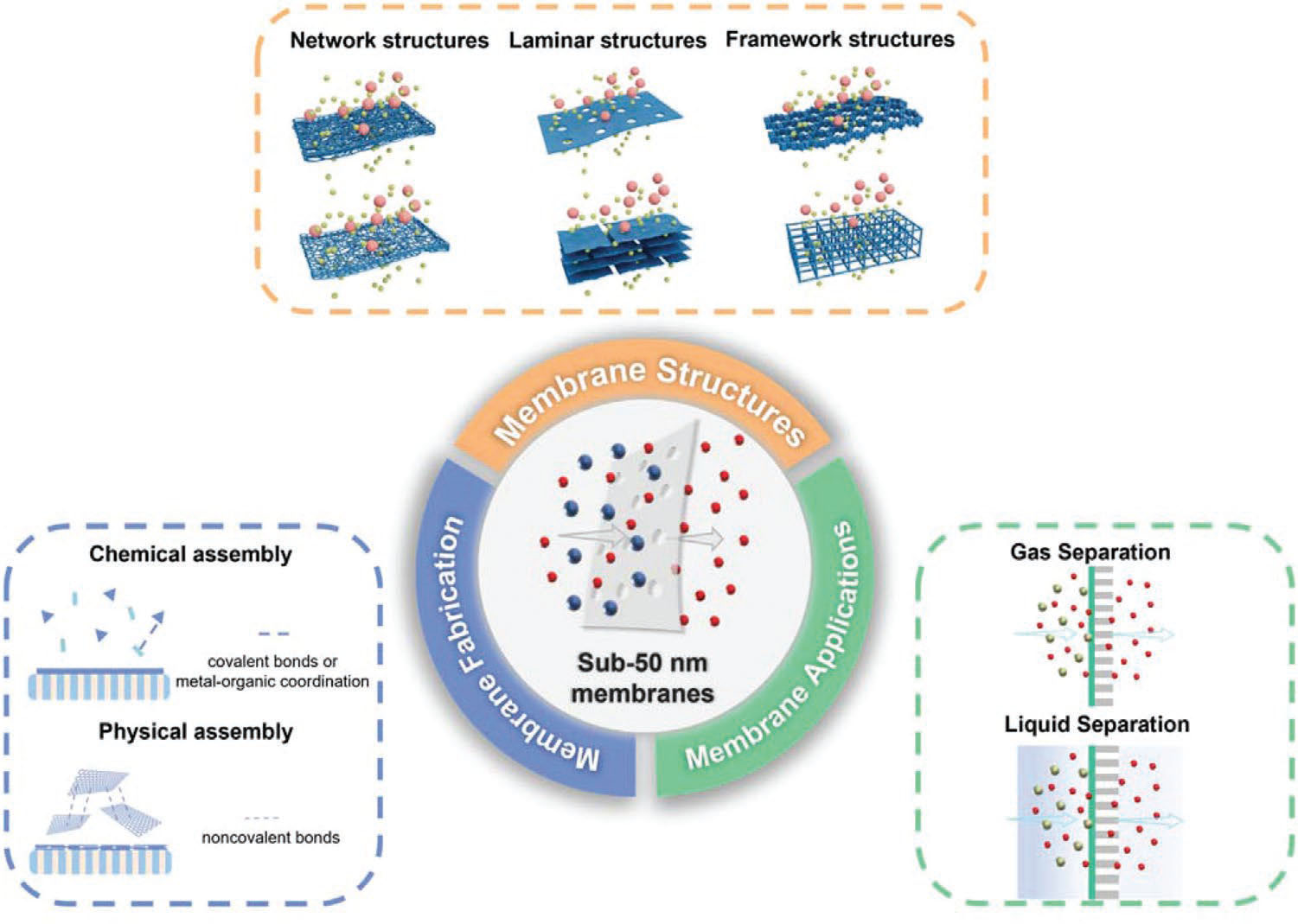 |
|
| An overview of membrane structure (network, laminar, and framework structure), fabrication methods, and applications of sub-50 nm membranes. (Reprinted with permission by Wiley-VCH Verlag) | |
| The review contains a detailed description of how new materials and advanced methods are developed to reduce membrane thickness and interesting mass transport phenomena across ultrathin layers, with attention to other factors concerning practical applications, that is, pore structure, stability and scalability. | |
| This is followed by an analysis and comparisons between their performances for industry-relevant commercial applications such as hydrogen separation, carbon capture, water purification, and desalination. | |
| The majority of sub-50 nm membranes that have been experimentally demonstrated so far in more than 100 publications include nanoporous atomic thin membranes (NATMs, ∼0.335 nm) based on graphene, graphene oxide (down to 1.8 nm), polyamide (<10 nm) membranes, metal-organic frameworks (MOFs), MXenes., covalent organic frameworks (COFs), and MoS2. | |
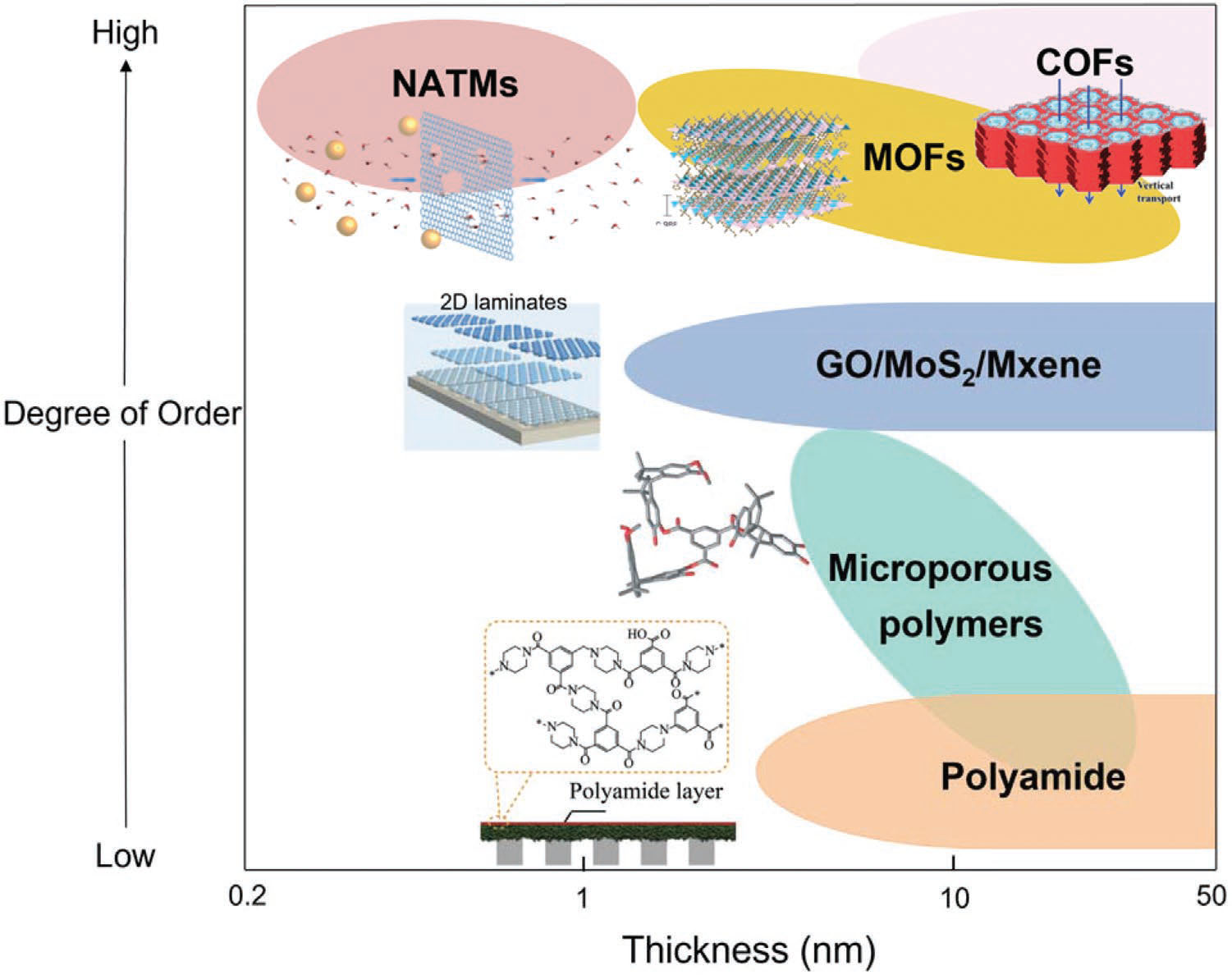 |
|
| Diagram of the thickness-degree of order relationship of some illustrative sub-50 nm porous membranes. (Reprinted with permission by Wiley-VCH Verlag) | |
| The fabrication methods of each ultrathin membrane vary from case to case, but they can be generally divided into two types: chemical assembly and physical assembly depending on the interactions among membrane building units. | |
| Chemical assembly, such as in situ growth and interfacial synthesis, refers to the process where the selective layer is directly formed on the surface of the support layer or at the interface of two immiscible solutions through covalent bonds or relatively strong noncovalent bonds like metal-coordination. | |
| Physical assembly implies that the selective layer, formed ex situ, is transferred, coated, or stacked onto the support layer through relatively weak intramolecular noncovalent bonds like van der Waals force and π-π stacking. These fabrication methods can be further subdivided into transfer, coating, and filtration. | |
| Layer-by-layer (LBL) assembly of chemical reactions between reagents also falls into this category. it may involve both covalent and non-covalent interactions and hence can fall into the categories of both chemical and physical assemblies. | |
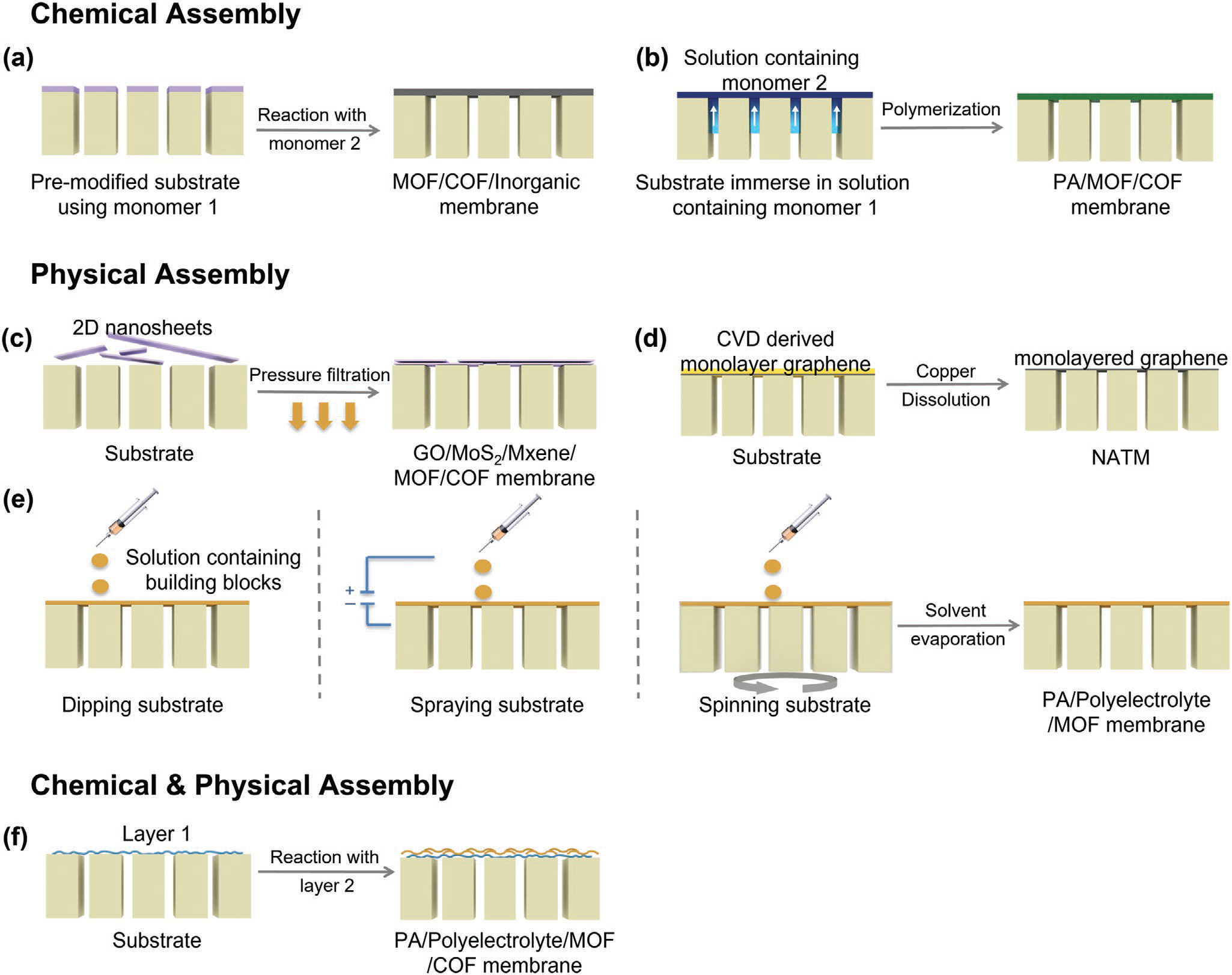 |
|
| General schematics for the fabrication methods of sub-50 nm membranes. a) In situ growth/biological adhesion; b) interfacial synthesis; c) filtration; d) transfer; e) coating and f) layer-by-layer assembly. (Reprinted with permission by Wiley-VCH Verlag) | |
| The final section of the review discusses the applications of sub-50 nm membranes in industry-relevant gas (H2 purification, CO2 capture) and liquid separations (reverse osmosis, nanofiltration) with a focus on commercial requirements. | |
| In their concluding remarks, the authors address various challenges and limitations that still need to be overcome, such as the complicated procedures and expensive raw materials that severely restricting their scale-up production; with regard to stability as a crucial concern in real-world operations, the long-term robustness of most ultrathin membranes need to be clearly demonstrated; ultrathin membranes must be examined in realistic separation conditions to reveal their suitable potentials for the process; and finally, future developments need to be guided by a fundamental understanding in mass transport at extremely low thickness. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
