| Jan 29, 2024 | |
New biocompatible nanotweezers enhance control over diverse nanoparticles with versatility and precision |
|
| (Nanowerk Spotlight) The precise manipulation of individual nanoparticles has long enticed researchers across disciplines like materials science, nanotechnology and biomedicine. But physics itself has imposed key barriers—namely the diffraction limit constraining conventional optical traps alongside insufficient laser intensities to induce stable trapping. This has left nanoscale tweezing an enduring challenge, lacking generalized platforms to trap the dizzying diversity of nanomaterials now central to cutting-edge applications. Each new particle material has necessitated specialized, tailored solutions dependent on bespoke fabrication strategies and customized setups incompatible with high experimental throughput. | |
| Now researchers at Shenzhen University have circumvented such limitations with the development of highly adaptable optothermal nanotweezers (HAONT). They reported their findings in Advanced Materials ("Highly-Adaptable Optothermal Nanotweezers for Trapping, Sorting, and Assembling across Diverse Nanoparticles"). | |
| Without surface modifications or convoluted electrode constructs, this versatile technique can manipulate single nanoparticles spanning just 3.3 to 200 nm across organic, inorganic and biological materials using remarkably low laser powers. | |
 |
|
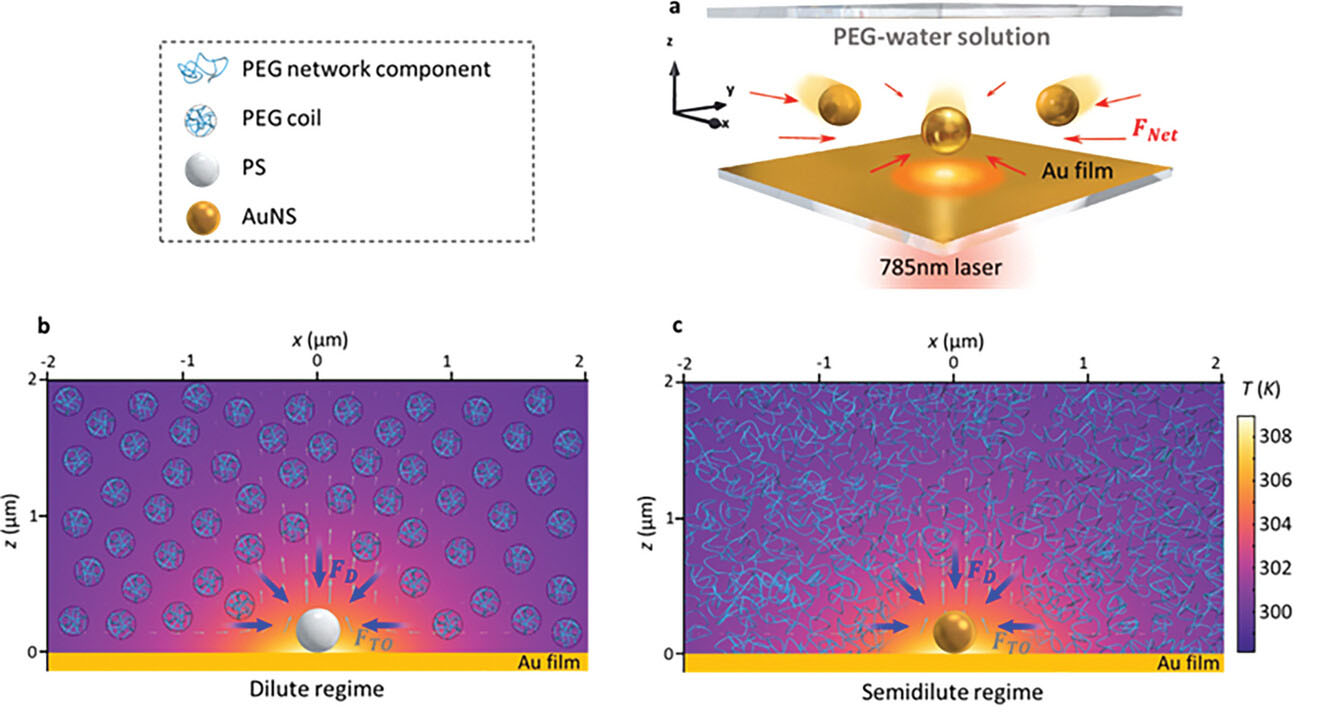
| The working principle of HAONT. a) The microfluidic channel consists of two cover glass slides, and the bottom one is coated with 10 nm Au film and a 5 nm chromium adhesion layer. The height of the chamber is 30 μm. The FNet is the net force for nanoparticle manipulation. b) Polystyrene sphere (PS) trapping in dilute PEG solution. c) Gold nanosphere (AuNS) trapping in semidilute PEG solution. The gray and blue arrows indicate the thermo-osmotic force (FTO) and diffusiophoretic force (FD) respectively. The incident 785 nm laser spot diameter is 1 µm, and the incident optical power is 0.3 mW. (Reprinted with permission by Wiley-VCH Verlag) | |
| Beyond nanofabrication and biosensing, HAONT’s biocompatibility, precision exceeding the diffraction limit and extensive nanoparticle compatibility helps crack longstanding barriers to nanoscale manipulation, unlocking new experimental possibilities from drug delivery insights to intercellular communication studies. | |
| Seminal work by Arthur Ashkin pioneering optical tweezers earned the 2018 Nobel Prize. But prevailing approaches still demand higher laser power densities, necessitating tailored solutions for specific nanoparticles. Moreover, unlike classic optical tweezers limited by diffraction barriers, HAONT operates beyond the diffraction limit, achieving remarkable precision down to sub-10 nm length scales. | |
| At HAONT’s core is an optothermal gold film inside a microfluidic chip that generates thermal gradients when hit by laser light. By tuning laser power and polyethylene glycol (PEG) polymer concentration in the surrounding liquid, researchers can manipulate two synergistic forces—diffusiophoresis and thermo-osmosis. The former relies on particle-polymer surface interactions to spur nanoparticle motions along thermal gradients. The latter utilizes fluid slip flows parallel to the gold-liquid interface caused by temperature differences. | |
| PEG is a known biocompatible surfactant, avoiding cytotoxicity issues plaguing other techniques. Unlike positive dielectrophoresis for instance, which requires electrode-laced setups, the electrode-free HAONT platform simplifies experimental configurations. Crucially, while ionic surfactants can deleteriously impact bio-nanoparticles, HAONT’s incorporation of nonionic PEG polymers circumvents this hurdle. | |
| Across organic and inorganic materials like polystyrene beads, gold nanoparticles and quantum dots sized anywhere from 5-200 nm, the researchers validated HAONT’s versatility. Using this technique, they assembled nanostars and printed specific nanoparticles onto cells, hinting at bio-sensing applications by enabling surface enhanced Raman spectroscopy. | |
| HAONT also facilitated nanoparticle sorting by tweezer stiffness and size. Intriguingly, when cranking up laser power, a doughnut vortex trapping scheme emerged that enabled novel cell-nanoparticle interplay. This doughnut configuration could significantly enhance the accuracy of nanoparticle-mediated drug delivery. | |
| Overall, the tool’s biocompatibility, nanoscale precision without surface changes and extensive manipulation range helps overcome limitations hobbling existing platforms. | |
| By accounting for the impact of a nanoparticle’s thermal conductivity relative to the surrounding liquid, researchers gained key insights into trapping behaviors. Metal nanoparticles with higher thermal conductivities than solutions homogenize thermal gradients, weakening net tweezering forces otherwise reliant on such gradients. The team found for example that 200 nm gold particles experienced nearly 60% lower net forces than similarly sized polystyrene beads. Smaller metal nanoparticles however saw more comparable tweezer stiffnesses relative to nonmetallic counterparts. Their computations revealed how a particle’s size and material governs the interplay between optical, diffusiophoretic and thermo-osmotic forces during HAONT manipulation. | |
| While promising, some constraints exist. HAONT lacks the accuracy of direct external force measurements possible with standard optical tweezers. The changing viscosity from thermophoretic polymer migrations also hampers precision. Moreover, manipulating single floppy biomolecules like DNA or proteins remains difficult given the technique’s reliance on particle-fluid interactions. And the required planar gold film prevents 3D tweezering. Despite such drawbacks, the tool’s ease of use, low optical power needs and applicability across diverse nanoparticle combinations bode well for adoption. | |
| With further enhancements like temperature regulation or applied electrical fields, HAONT could grow into a versatile nano-manipulation platform for synthetic biology, colloidal engineering, nanomedicine and materials science applications. | |
 By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
By
Michael
Berger
– Michael is author of three books by the Royal Society of Chemistry:
Nano-Society: Pushing the Boundaries of Technology,
Nanotechnology: The Future is Tiny, and
Nanoengineering: The Skills and Tools Making Technology Invisible
Copyright ©
Nanowerk LLC
|
|
|
Become a Spotlight guest author! Join our large and growing group of guest contributors. Have you just published a scientific paper or have other exciting developments to share with the nanotechnology community? Here is how to publish on nanowerk.com. |
|
