Measuring pH locally with terahertz spectroscopy
 Scientists have developed a new method to determine a local pH value near a specific site of a biomolecule.
Scientists have developed a new method to determine a local pH value near a specific site of a biomolecule.
Nov 20th, 2020
Read more
 Subscribe to our Biotechnology News feed
Subscribe to our Biotechnology News feed
 Scientists have developed a new method to determine a local pH value near a specific site of a biomolecule.
Scientists have developed a new method to determine a local pH value near a specific site of a biomolecule.
Nov 20th, 2020
Read more Skin and cartilage are both strong and flexible - properties that are hard to replicate in artificial materials. But a new fabrication process brings lifelike synthetic polymers a step closer.
Skin and cartilage are both strong and flexible - properties that are hard to replicate in artificial materials. But a new fabrication process brings lifelike synthetic polymers a step closer.
Nov 19th, 2020
Read more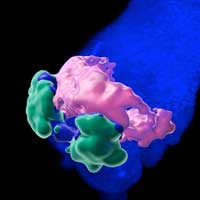 Bioengineers have used organoids - tiny lab-grown organs - to mimic the early development of the heart in the mouse embryo. The work is another step towards future bioartificial organs for research and transplants.
Bioengineers have used organoids - tiny lab-grown organs - to mimic the early development of the heart in the mouse embryo. The work is another step towards future bioartificial organs for research and transplants.
Nov 11th, 2020
Read more A new approach to modifying microbes' metabolic processes will speed up production of innovative bio-based fuels, materials, and chemicals.
A new approach to modifying microbes' metabolic processes will speed up production of innovative bio-based fuels, materials, and chemicals.
Nov 10th, 2020
Read more The method currently used to produce stem cell-derived tissues has a very limited throughput. By semi-automating tissue differentiation, researchers have made the process nearly four times faster, without compromising on quality.
The method currently used to produce stem cell-derived tissues has a very limited throughput. By semi-automating tissue differentiation, researchers have made the process nearly four times faster, without compromising on quality.
Nov 10th, 2020
Read more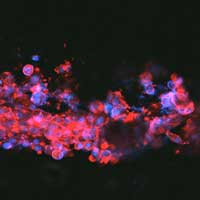 Researchers have developed a neuron-growing ink that uses the body's own electrical signals to precisely guide the growth of nerve cells.
Researchers have developed a neuron-growing ink that uses the body's own electrical signals to precisely guide the growth of nerve cells.
Nov 10th, 2020
Read more An interdisciplinary team of researchers present Biomedisa, an easy-to-use open-source online platform for biomedical image segmentation. The work addresses the needs of scientists without substantial computational expertise.
An interdisciplinary team of researchers present Biomedisa, an easy-to-use open-source online platform for biomedical image segmentation. The work addresses the needs of scientists without substantial computational expertise.
Nov 9th, 2020
Read more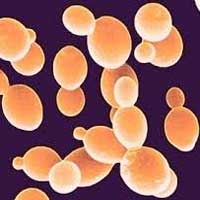 Scientists have found a way to use waste products as yeast feed. This exciting discovery opens up the possibility of fuelling biofactories without devoting arable land to producing sugars for them, but also because of the larger role methylotrophic organisms play in the environment.
Scientists have found a way to use waste products as yeast feed. This exciting discovery opens up the possibility of fuelling biofactories without devoting arable land to producing sugars for them, but also because of the larger role methylotrophic organisms play in the environment.
Nov 5th, 2020
Read more Scientists devised a method to turn shells from prawns and crabs into L-DOPA, a widely used drug to treat Parkinson's disease. A similar method can be used to convert wood waste to Proline, which is essential for the formation of healthy collagen and cartilage.
Scientists devised a method to turn shells from prawns and crabs into L-DOPA, a widely used drug to treat Parkinson's disease. A similar method can be used to convert wood waste to Proline, which is essential for the formation of healthy collagen and cartilage.
Oct 29th, 2020
Read more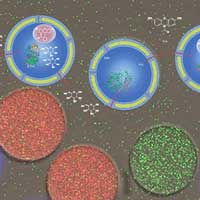 Scientists have developed a precisely controllable system for mimicking biochemical reaction cascades in cells. Using microfluidic technology, they produce miniature polymeric reaction containers equipped with the desired properties.
Scientists have developed a precisely controllable system for mimicking biochemical reaction cascades in cells. Using microfluidic technology, they produce miniature polymeric reaction containers equipped with the desired properties.
Oct 28th, 2020
Read more Researchers synthesize an elastic polymer from low-cost, sustainable materials, that can be used it to fabricate lenses that help keep infrared cameras focused in the dark.
Researchers synthesize an elastic polymer from low-cost, sustainable materials, that can be used it to fabricate lenses that help keep infrared cameras focused in the dark.
Oct 27th, 2020
Read more New genetic sensors could function as a lab test device and even as a live monitoring system inside living cells.
New genetic sensors could function as a lab test device and even as a live monitoring system inside living cells.
Oct 26th, 2020
Read more Cyanobacteria are among the oldest forms of life and are of great ecological importance. Genome sequencing will help to unlock the genetic potential of unusual cyanobacteria for the production of new active agents and to explore the potential for applications in biotechnology.
Cyanobacteria are among the oldest forms of life and are of great ecological importance. Genome sequencing will help to unlock the genetic potential of unusual cyanobacteria for the production of new active agents and to explore the potential for applications in biotechnology.
Oct 23rd, 2020
Read more Superior coating performance achieved compared to existing clinical products while reducing production cost and time.
Superior coating performance achieved compared to existing clinical products while reducing production cost and time.
Oct 23rd, 2020
Read more A new discovery may provide the crucial link that helps accelerate development of microalgae into beneficial human health supplements.
A new discovery may provide the crucial link that helps accelerate development of microalgae into beneficial human health supplements.
Oct 20th, 2020
Read more Using a magnetic field and hydrogels, researchers have demonstrated a new possible way to rebuild complex body tissues, which could result in more lasting fixes to common injuries, such as cartilage degeneration.
Using a magnetic field and hydrogels, researchers have demonstrated a new possible way to rebuild complex body tissues, which could result in more lasting fixes to common injuries, such as cartilage degeneration.
Oct 19th, 2020
Read more A new process uses pineapple leaf fibres to create ultra-light, biodegradable aerogels. They are effective as oil absorbents and for heat and sound insulation. They also have potential applications in food preservation and wastewater treatment.
A new process uses pineapple leaf fibres to create ultra-light, biodegradable aerogels. They are effective as oil absorbents and for heat and sound insulation. They also have potential applications in food preservation and wastewater treatment.
Oct 14th, 2020
Read more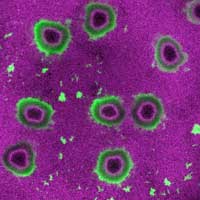 Studying bacterial biofilms, scientists have discovered that mechanical forces within them are sufficient to deform the soft material they grow on, e.g. biological tissues, suggesting a 'mechanical' mode of bacterial infection.
Studying bacterial biofilms, scientists have discovered that mechanical forces within them are sufficient to deform the soft material they grow on, e.g. biological tissues, suggesting a 'mechanical' mode of bacterial infection.
Oct 13th, 2020
Read more