| Posted: April 8, 2009 |
Silver chloride microparticles act as light-driven micromotors that organize into swarms |
|
(Nanowerk News) A swarm of tiny machines, speeding in concert through the bloodstream to repair an organ or deliver a drug to its target area, microrobots working together to construct a nanotechnological component—although it sounds like science fiction, it is a thoroughly realistic future scenario. Amazing progress has already been made in the production of autonomous nano- and micromotors, but the little machines have continued to lack in team spirit. To complete challenging tasks, the individual machines must communicate and cooperate with each other. Researchers led by Ayusman Sen at Pennsylvania State University (USA) have now introduced silver chloride microparticles that can “swarm” together, almost like living single-celled organisms.
|
|
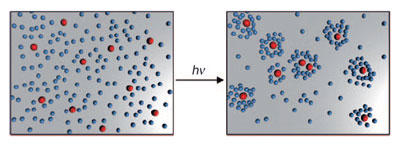
As reported in the journal Angewandte Chemie, irradiation with UV light causes the particles to give off “signal substances” that “attract” other particles (Schooling Behavior of Light-Powered Autonomous Micromotors in Water).
|
 |
| 0.004% wt. suspension of octadecylamine-modified nanodiamond demonstrates bright blue fluorescence under UV light, similar to that of selected natural diamonds. (Image: Yury Gogotsi, Drexel University)
|
|
Living cells and organisms are able to exchange information with each other to accomplish tasks as a team. Single-celled slime molds, for example, living in unfavorable conditions thus release a special substance. Neighboring slime molds follow the gradient of this signal substance and aggregate in the form of a multi-celled fruiting body. The silver chloride particles used by Sen’s team, which are about 1µm in size, behave in a similar fashion when irradiated with UV light. Silver chloride decomposes under UV light, releasing ions that act as both a propulsion mechanism and signal substance.
|
|
This phenomenon is based on diffusiophoresis, the movement of particles along an electrolyte gradient. The silver chloride particles “swim” toward a higher ion concentration. Because of irregularities in the surfaces of the particles and non-uniform irradiation, the degradation of the particles is asymmetric. Different quantities of ions are released in different places on the surface, which results in a local ion gradient around the particles. The particle thus produces its own ion gradient, which propels it at speeds up to 100 µm/s (self-diffusiophoresis). Neighboring sliver chloride particles follow the ion gradient of the solution and “swim” to regions of higher particle density. After several minutes, this results in small, stable “swarms” of particles. Photochemically inactive silicon dioxide particles also react to the ion signal, aggregating around the silver chloride particles.
|
|
This system can be used as a nonbiological model for communication between cells. Most importantly though, it represents a new design principle for “intelligent” synthetic nano- or micromachines that can work together as a team.
|

